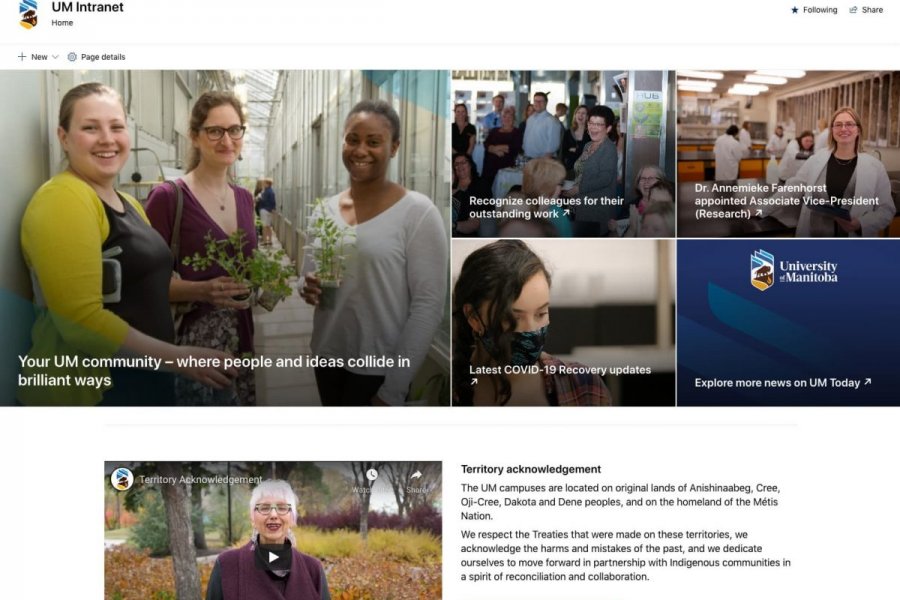
About UM Intranet
UM Intranet serves as your portal to conveniently and quickly access faculty and staff resources and materials needed for your career, life and work at UM.
Accessible only to those with a @umanitoba.ca email address (faculty, staff, nil appointments and affiliated partner employees), UM Intranet is hosted on SharePoint Online (SPO), a Microsoft 365 application commonly used for intranet sites.
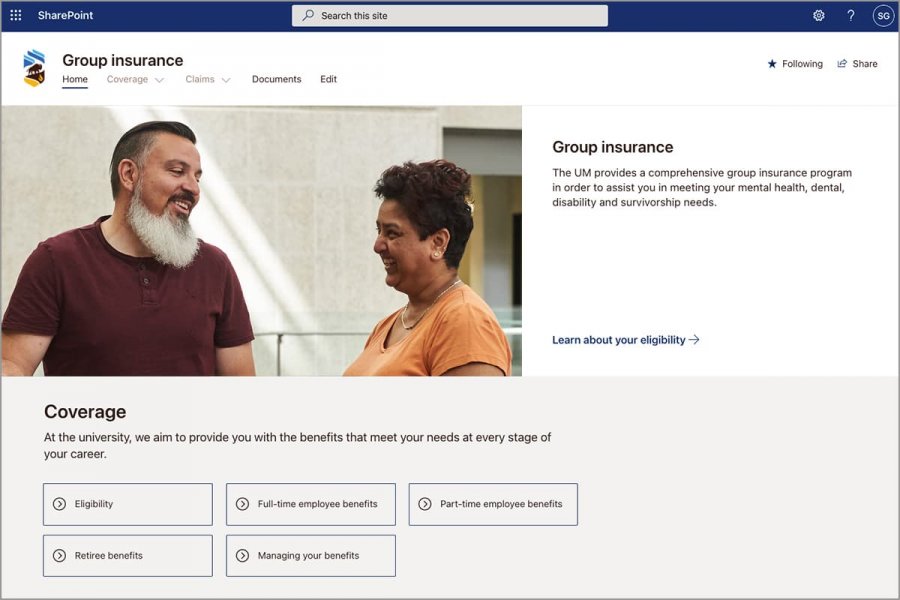
Employee resources and supports are grouped into five areas: Employee resources; Work supports; Campus; Units; and Apps and sites. Find out more about content categories on the Faculty and Staff webpage.
Learn more about UM Intranet content, navigation and supports through the information and UM Today stories below.
How do I find the content I need?
If you are a UM employee, nil appointee or affiliated partner with an active @umanitoba.ca email address, you can:
- Access through Faculty and Staff webpage on umanitoba.ca.
- Clicking on the UM Intranet button will direct you to log in to Microsoft 365 using your @umanitoba.ca email address and password; or
- if you are already signed into M365, you will go directly to the UM Intranet homepage.
- Find important information about content categories.
- Bookmark the Intranet homepage and access that way.
- You can bookmark your top pages for convenient future access.
- For the bookmarks to work, must be logged in to Microsoft 365 using your @umanitoba.ca email address and password.
- Previously bookmarked public website content will redirect to this Transition Page.
- Access from the SharePoint Online Dashboard if you are more familiar with the Microsoft 365 SharePoint Online application.
- Go to the UM Intranet Support site for a step-by-step guide to accessing the SharePoint online dashboard and how to follow your favourite sites. (You must be logged in for the support site link to work.)
Support
Logging in to M365
To access UM Intranet, you will be prompted to log in to Microsoft Office. You must enter your @umanitoba.ca email address and password.
Visit the Microsoft support site to learn more about ‘How sign in works in Microsoft 365’.
I tried to log in, but received an Access Denied notification
Did you use a @myumanitoba.ca email or an external (non umanitoba.ca) email address? Try again using your @umanitoba.ca email address.
If you attempted to log in using your @umanitoba.ca email address, please contact the IST service desk at servicedesk@umanitoba.ca for further assistance.
I tried to log in and my password is not working/my credentials don’t work
Are you using your @umanitoba.ca email address to log in to UM Intranet? If not, try again with the proper credentials
Are you already signed in to Microsoft 365 using an email address from another organization?
- Option 1: Try opening a private or incognito window in your browser. In the new window, copy and paste the URL you are attempting to access. On the Microsoft login screen, use your @umanitoba.ca email address and password.
- Option 2: Sign out of Microsoft 365. Sign in again by selecting ‘use another account’ and entering your @umanitoba.ca email address and password.
I don’t know if I have a @umanitoba.ca email address
If you are an employee of UM:
- Go to Sign UM and claim your ID.
If you are a nil appointment or affiliated partner (primary employer is not UM):
- You will be given an employee number and you can claim your ID.
I have to log in to Microsoft 365 multiple times a day
This may be because of your browser cookies settings. Please contact IST Service Desk at servicedesk@umanitoba.ca for support.
Which browsers work with Office for the web?
You can find out more information on the Microsoft support site for a comprehensive breakdown of the recommended browsers for your device.
If you are having trouble accessing UM Intranet, submit an IST service request to servicedesk@umanitoba.ca.
If you are having trouble finding content on UM Intranet, contact the respective content owner of the content (i.e., HR Service Desk, Registrar’s Office, or the Faculty).
Our faculty/unit would like to have an intranet site. Who do I contact?
Check with your unit or faculty communications specialist. They can connect with their Strategic Communications contact to determine whether an employee-only site is right for your content.