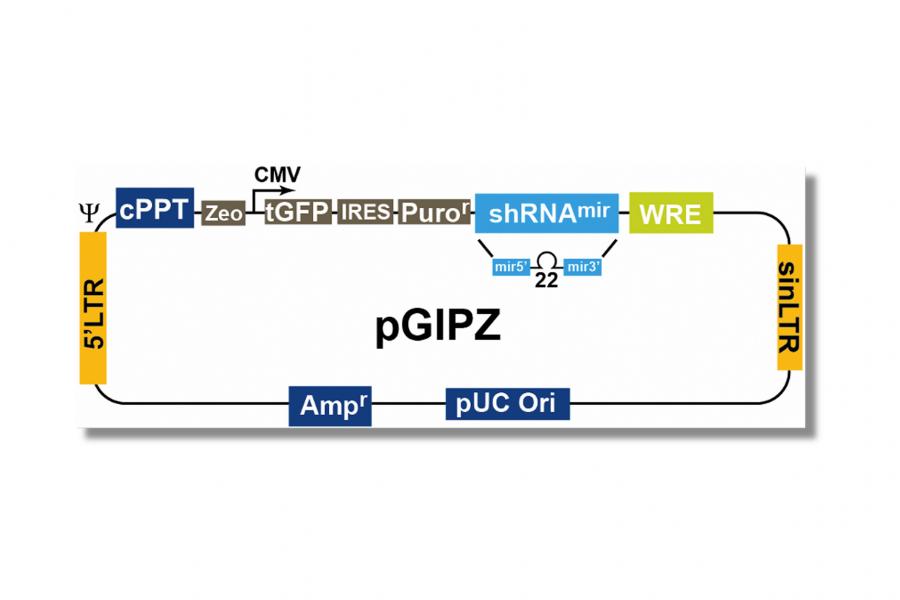
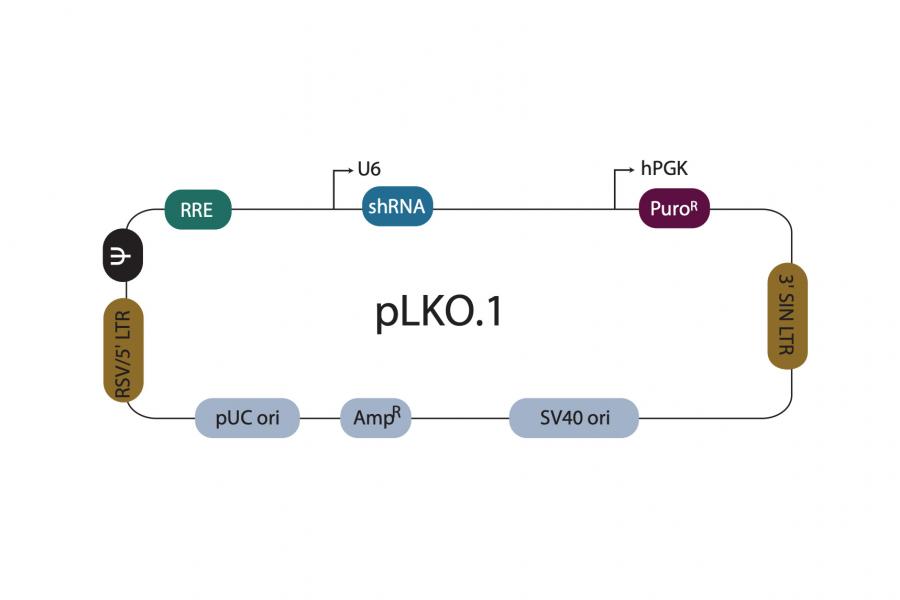
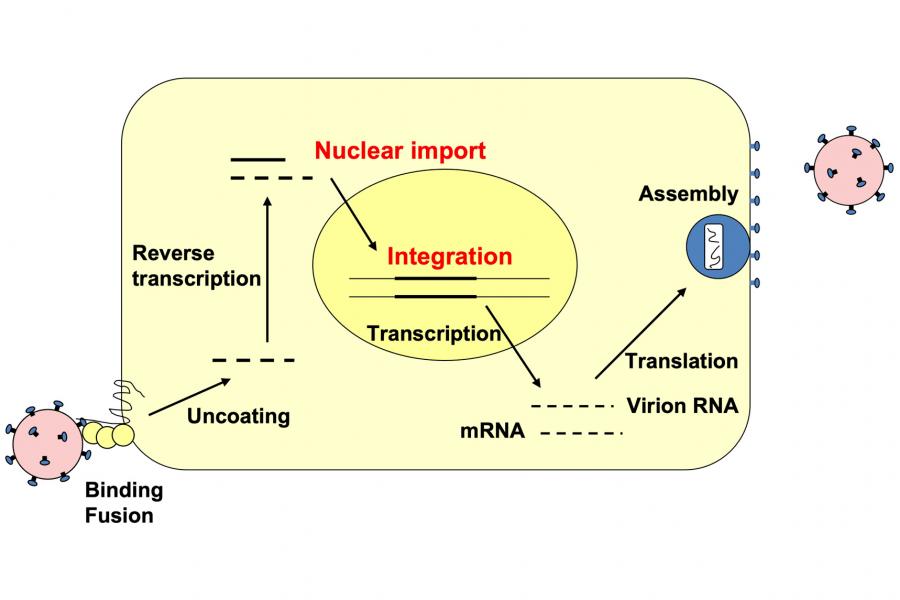
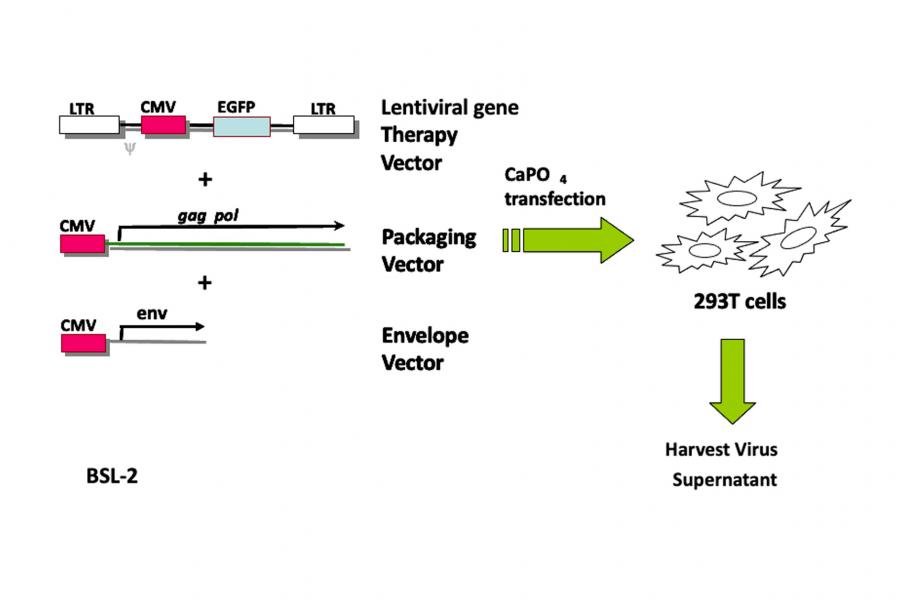
Preparation:
Change medium of suspension cells one day before the experiment to ensure log phase growth.
Infection:
On the day of infection, collect cells and count them.
Aliquot 1x105 cells per capped centrifuge tube and spin at 4000 rpm for 2 minutes.
Remove the supernatant and add 250 μl of virus supernatant appropriately diluted (refer to the ‘virus preparation’ protocol). Mix by pipetting.
Incubate for 2 hours at 37°C in an incubator.
After 2 hours, add 1 ml of fresh medium.
Pellet cells as in step 2, and remove the supernatant.
Add 0.5 ml of fresh medium to each tube and transfer the cells to a 24-well plate.
Analyze the infected cells after 72 hours.
If puromycin selection is required:
After 72 hours, remove the medium from the well and transfer it to a tube. Add 500 μl of fresh medium to the wells. Spin the tube at 1200 rpm for 5 minutes and resuspend the pellet in 500 μl of medium containing puromycin. Put these 500 μl back to the well.
On the next day, remove the medium from the well that is undergoing puromycin selection and add 500 μl of fresh medium.
Alternatively, you can follow this protocol:
Do a cell count (0.1x106 cells is best for transduction).
Add cells to an Eppendorf tube.
Centrifuge cells at 2000 rpm for 5 minutes.
Remove supernatant and resuspend pellet with 250 μl of virus suspension.
Put the solution in a 24-well plate.
Centrifuge at 2000 rpm for 1 hour at 24°C.
Remove supernatant (place pipette tip at the corner of the well and draw up liquid very slowly).
Resuspend pellet in 0.5 ml of cell culture medium.
Incubate at 37°C with 5% CO2.
Note that the concentration and timing of puromycin selection may vary from cell to cell.