
The Manitoba Population Research Data Repository
What is the data repository for?
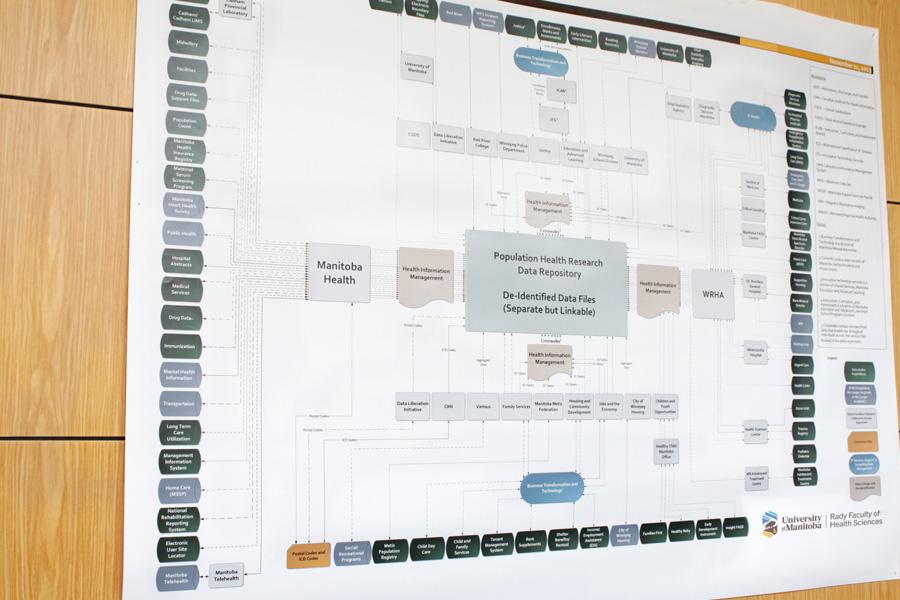
The data repository is a resource for researchers wishing to study the health and social well-being of Manitobans.
The data come from a variety of government department administrative datasets, such as:
Health care
Visits to hospitals, physicians, emergency departments and specialists, homecare, and personal care homes, pharmaceutical prescriptions, and COVID-19 (vaccinations, lab testing and results, and surveillance) data.
Education
Public school and University of Manitoba classes, marks/grades, graduation rates, programs to support kids and adult training programs to support employment.
Social / Families
Services provided to support families, children in care, employment/income assistance and social housing.
Justice
Prosecutions, court appearances, and charges/outcomes at disposition.
Registries
The Manitoba Health Insurance Registry, Vital Statistics Mortality Registry, Immigration Permanent Residence Database, Manitoba First Nations Research File, and Métis Population Database.
The data are de-identified, so that we can track individual interactions with services, but cannot identify the people seeking them.
Data list and data descriptions
Applying for access and use
To use the data repository for research you must:
- complete an accreditation session
- submit a research proposal and project feasibility and data access quote request form,
- if your research project will use "research collected" (project) data (e.g. survey, RCT), please contact the Centre for Healthcare Innovation (CHI) before completing your proposal
- if your research project will involve multiple Canadian provinces, please contact the Health Data Research Network (HDRN) Canada - Data Access Support Hub (DASH) before completing your proposal
- confirm project funding
- obtain approval from the Provincial Health Research Privacy Committee (PHRPC), University of Manitoba (UM) Health Research Ethics Board (HREB) and (if necessary) other data providers
- sign a researcher agreement and submit approval documentation
Each step is outlined below.
Step 1: Attend accreditation session
Overview
Accreditation is required for:
- all those with access to a Manitoba Centre for Health Policy (MCHP) computer system
- all principal investigators on projects being carried out at MCHP
Your first accreditation session must be a live session, conducted either in person or via teleconference. Subsequent accreditation sessions must be completed annually and verified online by completing an annual renewal questionnaire.
Resources
Accreditation - updates for 2024 (pdf)
Annual accreditation renewal sessions must be verified by completing an online accreditation renewal questionnaire.
Contact
For more information on data access, use, and accreditation, contact the Accreditation Coordinator.
Step 2: Research proposal
Overview
Your research plan must contain a research proposal as well as a list of all of the databases you plan on using in your project.
Tasks
Develop your research proposal
The proposal describes the objectives, methods and resource requirements for your research project in as much detail as possible.
Currently, there is no template for writing the proposal. However, we do recommend using the general structure described in the Proposal format section.
- if your research project will use "research collected" (project) data (e.g. survey, RCT), please contact the Centre for Healthcare Innovation (CHI) before completing your proposal
- if your research project will involve multiple Canadian provinces, please contact the Health Data Research Network (HDRN) Canada - Data Access Support Hub (DASH) before completing your proposal
Your overall research plan must also include a list of the databases you will use in your research project. Please see the next step in the applying for access and use process for more information on identifying the databases you want to use in your research.
Additional tasks for students
Find a Manitoba Centre for Health Policy (MCHP) affiliated researcher who will sit on your thesis committee.
NOTE: There are specific requirements for this type of arrangement. Please review the Guidelines for student use of the repository (PDF) for more detailed information.
Meet with your proposed supervisor and the MCHP Associate Director, Repository, Data Access and Use to discuss the proposed research.
Proposal format
The research proposal is a detailed description of your research project and is an integral piece of the research plan. It is written by the primary investigator / project team and should contain the following information:
A. Research objectives
A list of objectives / purpose of the research and a description of what your research project is trying to achieve.
B. Methodology section
As a minimum, please include the following information:
Type of study
Describe the type of study you are proposing:
- Identify if the study design is cross-sectional or longitudinal
- If a cohort is required, please describe the cohort in as much detail as possible
- If the study is longitudinal, changes in data values occurring over time need to be taken into account. Examples of this include residential moves / changes, classification / coding changes (e.g. Hospital Abstract coding change from ICD-9-CM to ICD-10-CA on April 1, 2004), and family structure changes / alterations.
For more information on the types of studies available, please read MCHP Study Design and Methods.
Type of analyses
Describe the analytical methods that you plan on using in your project.
- Indicate whether existing Manitoba Centre for Health Policy (MCHP) methods / techniques are to be used, or if new methods need to be developed. The MCHP Concept Dictionary and Glossary contains information on many different methods already developed by MCHP using data in the Repository. Examples include income quintiles, geographical analysis by health regions, and the Charlson Comorbidity Index.
- If the methods/analytical techniques are new, please describe them or provide references to available information.
- If possible, indicate whether original data fields can be used or whether values must be derived. For example, if you are creating a scale of values, indicate the categories and what range of values fit into each category.
Identification of disease / conditions / indicators / outcomes / dependent variables
Describe the specific diagnoses / conditions, procedures / interventions, and the indicators and outcomes that will be investigated in your project:
- If you are defining specific diseases, conditions, medications or procedures, please specify the codes you plan to use in your study, if you know them. This may include specific International Classification of Diseases (ICD) codes using ICD-9-CM, ICD-10, or ICD-10-CA and the Canadian Classification of Health Intervention (CCI) codes for diagnoses and procedures/interventions; physician tariff codes for services provided; or drug identification numbers (DINs) for identifying prescriptions.
- MCHP has developed many specific indicators / measures related to the areas of health, social, education and justice that can be used for different types of analyses. Examples include: premature mortality rate, children in care, high school graduation, and involvement in the justice system.
- The MCHP Concept Dictionary and Glossary contains information on the codes used in MCHP research to define many different diagnoses, conditions and procedures / interventions, as well as algorithms for defining different types of indicators. This information will assist in identifying the databases you will need to use in your research.
NOTE: The use of multiple codes within various time frames (as opposed to a code at one point in time) may require extra programming time.
C. Project location
Please specify where your programming / analyses will take place. Indicate if you are planning to work with an MCHP analyst or through remote access.
Please see the Remote access site - user and site process (PDF) for additional information.
NOTE: Under the existing data sharing agreements, MCHP cannot provide extracted data subsets outside of MCHP's secure environment.
D. MCHP resources
Please indicate the type and level of support required from MCHP for your research project. MCHP will provide data and analytic support on a cost recovery basis, with timelines dependent on the availability of MCHP resources.
The types of support you may require include:
- Data extraction - MCHP will extract the necessary data from the Repository for your project and provide you access to it in a secure environment.
- Programming / data analyses support - if required - do you have your own programmer/data analyst resources or will you require MCHP to provide programming/analytic support for your research project? Are basic descriptive analyses, such as frequencies and means, sufficient for completion of analyses, or are more complex analyses like hierarchical linear modelling, exploration of interaction effects necessary? MCHP will provide resources for SAS programming code development and data analyses support for your project on a cost recovery basis.
- Physical / computer workspace setup - if you require physical / computer workspace at MCHP please indicate this in your proposal.
E. Additional resources
If your research project has additional resource requirements, please describe them.
This may include:
1) Using project specific data that is not currently available in the data repository. Using project specific data involves additional steps and costs to your project, and requires written approval, such as a Data Sharing Agreement (DSA), from the organization providing the data.
The steps involved in using project specific data in your research project include:
- creating a DSA with the organization providing the project specific data
- data linkage at Manitoba Health (MH)
- a transfer of "linkage / crosswalk" data between MH and MCHP
- a transfer of service data between the data provider and MCHP
- data installation at MCHP
- time for MCHP to learn more about the project specific data (if required)
For more information on using project specific data in your research project, please read the Guidelines for requests for project specific data (PDF) and the information in Step 3 of the applying for access and use section.
2) Involvement/collaboration with the George and Fay Yee Centre for Healthcare Innovation (CHI) - services provided by CHI include clinical research and methods, data analytics and management (e.g. use of REDCap for collecting and storing project specific data), a learning health system, patient and public engagement, and patient reported measurement.
For more information, see CHI Services or contact CHI.
To request a free consultation, please complete and submit a:
CHI and MCHP Request for Service form
3) Involvement/collaboration with the Health Data Research Network (HDRN) Canada - for multi-jurisdictional (e.g., multi-provincial) research projects.
For more information, see:
F. Recommendations and additional guidelines
We recommend that an MCHP researcher familiar with the applying for access and use process and the use of the Data Repository be included in your research project team.
For students - if the proposal is for a student thesis, please review Guidelines for student use of the repository (PDF). Graduate students (PhD and master's level) are expected to submit their research plan separately from any ongoing research project. In most cases, resident and undergraduate (e.g.,: BSc Med) research projects must be done as part of an existing, approved research project of an advisor.
For private sector / industry sponsored research – research projects seeking or acquiring private sector or industry sponsored funding have additional rules and guidelines. For more information, please review the following information:
Contact
Contact Charles Burchill for assistance prior to developing your research proposal.
Step 3: MCHP project feasibility and data access quote request form
Overview
The Manitoba Centre for Health Policy (MCHP) Project Feasibility and Data Access Quote Request form is used to collect information about your research project and identify all of the databases you want to use in your research project.
The entire research plan, the research proposal and the completed form, must be submitted to MCHP for feasibility review.
The form is available in two versions:
About the form
The MCHP Project Feasibility and Data Access Quote Request form contains four sections and two appendices that collect the following information:
A. Project title and principal investigator
- the full title of the project
- the name of the principal investigator and contact information (e-mail address, institution/department, telephone number, and full mailing address)
- the project coordinator or principal investigator's assistant (name and email address)
- an indication whether the project is a thesis. If it is, indicate the level or type of thesis and the thesis supervisor's name and email address (see the Guidelines for student use of the repository (PDF) for more information)
- the name of the University of Manitoba project associated investigator (if required)
B. Project Information
Project description
This can be the Research Proposal developed in Step 2 or a letter of intent
Whether the project is related to another MCHP project
If it is, add the project reference number(s)
Data requirements
Indicate Appendix 1 for Repository data. Indicate Appendix 2 if your project also uses project specific data that is not available from the Repository. See below for additional information on completing Appendix 1 and 2.
NOTE: Using non-repository data, or project specific data, for your research project requires additional steps in the MCHP data access process.
See Guidelines for requests for project specific data (PDF) for additional information on this process.
Whether your analyses will include any specific population subgroups
This could be First Nations, Metis or Inuit but includes any that require privacy considerations/permissions for use.
Where the data access / analyses will be located
At MCHP or through remote access. (For more information on remote access, view the Remote access site - user and site process (PDF)
C. Funding information
- indicate the source of funding, if known
- if funding is being applied for, please indicate this on the form
- the name of the PI on the funding submission
- the amount expected/received
- the expected project start and end dates
D. MCHP accreditation status
The researcher's MCHP accreditation status within the last 12 months.
NOTE: MCHP accreditation must be completed prior to the start of a research project and must be renewed on an annual basis. For more information, please see Step 1 - Attend Accreditation Session.
Appendix 1. List of databases required
A list of data repository databases required for your research project is necessary for a feasibility review, and the database list is also required for submission for project approvals. The information should be presented in table format with the following column headings:
- Data provider/type - this indicates the type of data (e.g.,: health, education, social, justice) you are requesting
- Database name - the name of the database
- Data years - the range of data years required for your project (e.g.,: 1995/96 to 2017/18)
- Data fields/variables - if known, list the data fields/variables and the indicators required for your project
- Rationale - the reasons for using the database in your research project
Resources available
The following resources are available to assist you in identifying relevant repository database information and the years of data available that you require for your project:
- Repository Data List - provides a summary list of databases available in the Repository, including the database name, years of data available, and the associated data provider
- Repository Data Descriptions - a searchable tool that provides more detailed information on the content available in each database
- Chart - available years of data (PDF) provides a graphic illustration of the years of data available for each database
- MCHP Concept Dictionary and Glossary - a searchable tool that can provide detailed information on the existing algorithms (e.g. data sources, data field/variable names, code values, time frames) that are used in defining diagnoses, medical conditions, procedures, and health and social measures/indicators (e.g. provider visits, premature mortality, income quintiles) you may want to use in your research
- See Example list of MCHP repository databases required for project (PDF) for more information
Appendix 2. Database not available in the MCHP Repository
NOTE: Completion of Appendix 2 is only required if you are also planning to use project specific data, that is not part of the Manitoba Population Research Data Repository. The project specific data must be identified and approved for use.
Appendix 2 has two sections to complete, including:
A. Database information
Full title of the project (this should be the same information as recorded in section A of the FORM)
Principal investigator's name
Database name (assigned to the project specific database)
Database source (the organization / department / group where the database originates from)
Contact person at source (the name, e-mail address and phone number for the contact)
Organization providing/approving use of the project specific data and assuming liability for disclosure and use (e.g. Privacy Agreement under the Personal Health Information Act (PHIA) trustee)
Database scope - a description of the data, such as, contains demographic and personal health information, start and end dates, a brief description of the content, and whether the data is provided at an individual person or aggregate level
Data sharing arrangements - indication of whether there is a data sharing agreement for ongoing use of the data, or if the data will only be used for this specific project
NOTE: For more information, review the Guidelines for requests for project specific data (PDF)
B. Requirements for the transfer and use of project specific data
A signature in this section is required from the principal investigator, who is responsible for arranging the collection and disclosure of project specific data to MCHP, including all the necessary approvals regarding privacy and ethics from the project specific data providers.
Important notes about your list of databases
The primary data approval bodies (both the Provincial Health Research Privacy Committee (PHRPC) and the University of Manitoba (UM) Health Research Ethics Board (HREB)), MCHP and other approval agencies require a list of all the data that will be used in your project.
Creating one table with the following information will meet the requirements of PHRPC, HREB, MCHP and other approval agencies: the data provider/type, database name, years of data required, data fields / variables required, and the rationale / explanation of how the requested data relates to the study purpose, question(s), and hypotheses.
If the data is not directly related, include an explanation of why the data is required.
Creating a table that includes the information identified above means you will only need to create one version of the table for your project that can be submitted to the various organizations for approval.
Tasks
A. Complete the project feasibility and data access quote request form
B. Submit your research plan for feasibility review
The research plan must include:
- Research proposal
- if your research project will use "research collected" (project) data (e.g. survey, RCT), please contact the Centre for Healthcare Innovation (CHI) before completing your proposal
- if your research project will involve multiple Canadian provinces, please contact the Health Data Research Network (HDRN) Canada - Data Access Support Hub (DASH) before completing your proposal
- Project feasibility and data access quote request form
- Any additional supporting documentation for the proposal such as funding support, grant proposals, etc.
The information can be sent via email to the MCHP Repository Access Unit.
Private sector/industry sponsored projects
Fees
A non-refundable fee of $2,000 is required to allow for review of the funding contract. An invoice will be sent from the Manitoba Centre for Health Policy (MCHP) finance team for payment. The fee is not an assurance that a project will receive approval. Additional fees may be applicable from other data providers / organizations (e.g., UM Health Research Ethics Board (HREB)) involved in private sector / industry sponsored research projects.
NOTE: Research projects funded by the public sector (e.g., the Government of Canada, Health Canada, Public Health Agency of Canada (PHAC), Province of Manitoba) are not subject to this fee.
Using project specific data for your research project involves additional steps and costs to your project, and requires written approval from the organization providing the data.
Steps
The steps involved in using project specific data in your research project include:
- developing a data sharing agreement (for ongoing use of the data) or a letter of approval for using the data for your project
- data linkage at Manitoba Health (MH)
- a transfer of "linkage/crosswalk" data between MH and MCHP
- a transfer of service data between the data provider and MCHP
- data installation at MCHP
- time for MCHP to learn more about the project specific data (if required)
Important: a copy of the funding contract/agreement must be submitted with your research plan.
More information
For more information on using project specific data in your research project, please read the Guidelines for requests for project specific data (PDF).
For more information about private sector/industry sponsored research, please review the following information:
For students, please see Guidelines for student use of the repository (PDF) for more information.
Step 4: Feasibility review and project funding confirmation
Overview
The feasibility review looks at the information in your research plan, including the research proposal and the MCHP project feasibility and data access quote form, to ensure your research project is possible.
Feasibility
The feasibility review of your research plan usually takes 10 working days.
If the project is not feasible or requires clarification, you will be contacted by MCHP to discuss alternative solutions or additional information that is required. This may extend the amount of time MCHP requires to review your research plan.
If the project is feasible, you will receive an email from the MCHP Repository Access Unit (RAU) that contains:
- a formal signed reply from the associate director of the repository confirming that the project is possible, including a "quote letter" estimating the costs of your project. The quote is valid for one year from the date of the letter.
- if necessary, a list of additions or modifications to your research plan. This may include: additional databases you should request for your project, a list of codes/values that can be used to identify specific diagnoses or medical conditions, or other suggestions
- a "Next Steps" document that outlines what needs to happen after you acquire funding for your project
NOTE
The amount of time from the initial submission of the MCHP project feasibility and data access quote form to receipt of all the necessary approvals and permissions will take approximately four months.
Tasks
The required tasks include:
Cost estimate received - MCHP will provide you with an estimate of the costs associated with your project (the "quote letter"), and additional suggestions that should be updated in your research proposal and/or the MCHP project feasibility and data access quote form. The cost estimate can be used for grant applications.
"Next Steps" document received - MCHP will also provide you with a document that outlines what needs to happen after you acquire funding for your project, and a list of the databases that can be used for the submission of approval documentation.
Update your research plan (if necessary).
Acquire funding - if you already have funding in place for your project, please send this information to the MCHP Repository Access Unit (RAU). If not, you will need to explore opportunities and acquire funding for your project.
Send confirmation of funding - send confirmation of funding to the RAU prior to moving on to the next step.
Funding
Financial supports and opportunities for students
There are a number of financial supports, awards and opportunities available for students. See the Supports , awards and prizes section of our website.
Funding confirmation
Project funding confirmation is required by MCHP prior to obtaining approvals and permissions. Please forward a copy of the funding confirmation to MCHP when you receive it.
Before you submit documentation for any approvals, you must have proof of funding in place for your research project.
Contact
For more information, contact the MCHP Repository Access Unit.
Step 5: Privacy, ethics and data provider approvals/permissions
Overview
Every research project must obtain approvals/permissions from the appropriate data providers/organizations before the research project can begin. The type and number of approvals/permissions required depends on who is doing the research, the type of data the project is planning to use, and the scope of the research being proposed.
NOTE
Starting January 1, 2022 all research projects must follow the process outlined by the Research Improvements Through Harmonization in Manitoba (RITHIM) initiative. For more information on RITHIM see the RITHIM website - https://www.rithim.ca.
The Provincial Health Research Privacy Committee (PHRPC) must "review all health research projects that request or require use of personal health information held by any Manitoba trustee, including government and government agencies, and render a decision (i.e., approved, conditionally approved or not approved/requires revision)." For more information on the PHRPC see the Provincial Health Research Privacy Committee (PHRPC) Site Map.
About approvals
Every research project must obtain approval from the Provincial Health Research Privacy Committee (PHRPC) and the University of Manitoba (UM) Health Research Ethics Board (HREB). The Principal Investigator (PI) is responsible for submitting and obtaining approvals from PHRPC and HREB.
Additional approvals/permissions are required if you are using data from non-Manitoba Health (non-MH) Data Providers. If data/analyses directly or indirectly identify First Nations, Métis, and/or Inuit populations then additional partnership and approvals will also be required. The MCHP Repository Access Unit (RAU) will assist you in submitting the appropriate documentation for approvals/permissions.
All approval submissions must identify the same PI, project title and data request table. Failure to do so may result in delays to your project.
For graduate students, you must have your research project approved by your committee prior to submitting any other approvals. Graduate students are expected to obtain your own set of approvals for your research project. For residents and undergrad students planning a project under an existing research project, no additional approvals are required. However, the existing research plan / proposal must be amended to include the student as part of the project personnel. For more information, see the Guidelines for student use of the repository (PDF).
Note: Please do not make amendments during the initial proposal and approval process. This will only result in delays to the approval process.
Approvals/permissions required
The approvals/permissions required include:
1. Provincial Health Research Privacy Committee (PHRPC)
See PHRPC Submission Information web page for current information about the PHRPC submission process, including relevant links to the following forms and guides:
- Does my project require PHRPC review?
- PHRPC Application form
- Guidelines for completing a PHRPC submission form
- Conflict of interest disclosure form
- PHRPC protocol amendment form
- Guidelines for submitting a HIPC/PHRPC protocol amendment
Additional information on this page provides links to:
- Requirements for the collection and use of PHINs for research purposes (pdf)
- information on submission deadlines
- submission requirements
The PHRPC Information for Researchers web page contains additional information on:
- HIPC applications that received conditional approval in 2021
- Amendments to research projects approved by the HIPC or PHRPC
- New research applications
- Contact information for further questions or submitting an application
2. University of Manitoba (UM) Health Research Ethics Board (HREB) Requirements
- HREB approvals can be obtained by all UM employees, students, academic appointments and researchers affiliated with the Winnipeg Regional Health Authority (WRHA). Submissions to HREB must include a UM or WRHA associated researcher.
- See the UM Office of Ethics and Compliance - Submission Requirements web page for current information and guidelines about the HREB submission process, including: submission tips, initial review requirements, meeting dates and submission deadlines, new submissions, protocol amendments or changes to the study, annual review procedures and additional information.
NOTE: Please check for updates to the PHRPC and HREB requirements before submitting your application as the information / process changes periodically.
Additional notes
The nature of most research using the MCHP Data Repository requires completion of the HREB Submission Form for Retrospective Chart or Records Review.
Obtaining approvals from PHRPC and HREB is the responsibility of the Principal Investigator (PI) or project coordinator.
PHRPC, HREB, and other approval submissions may be submitted at the same time, but processing time may vary depending upon approval committee meeting schedules.
Your project may be subject to review by the University of Manitoba Research Quality Management Office.
Non-UM researchers and external researchers may require confirmation from other ethics boards that the proposed research project meets the required ethical standards.
NOTE: Depending on the scope of your research project, additional approvals/permissions may be required from:
3. Non-Manitoba Health (Non-MH) Data Providers (if applicable)
If your project requires access to data from non-MH data providers (e.g., WRHA, Education, Family Services, Justice) you must submit additional approval requests for use of the data and receive approvals from these agencies.
The required approval agencies will be identified in the "quotation" letter from the MCHP Associate Director of Research Access and Use.
The MCHP Repository Access Unit (RAU) will provide you with the necessary templates or processes for requesting approvals from non-MH data providers after you confirm that you have acquired funding for your project. Please do not use templates from prior submissions as these processes may have changed.
4. First Nations, Métis, and/or Inuit Representatives (if applicable)
MCHP is committed to partnership and collaboration when working on research projects that directly or indirectly identify First Nations, Métis, and/or Inuit populations as part of the research carried out using the Manitoba Population Research Data Repository.
We have made a commitment when using data that directly identifies Indigenous populations, or a primary outcome could be attributed to an Indigenous population, that partnership and collaboration will be sought from the appropriate individuals, communities, or representative groups. MCHP is committed to research following First Nations/Métis/Inuit ethical standards.
Partnership and collaboration will be sought when:
First Nation, Métis, or Inuit individuals or communities will be identified specifically
This includes research that specifically looks at whether a participant is First Nations, Métis, or Inuit (e.g., use of the Manitoba First Nations Research File or Métis Registry, or use of a field that identifies participants belonging to a specific First Nations, Métis, or Inuit group) and the research results use First Nations, Métis, or Inuit community membership as a specific variable, or the interpretation of the results refer to First Nations, Metis or Inuit language or culture.
This would include the use of data that is collected through specific authorities or groups that represent Indigenous populations (e.g., Child and Family Services (CFS) authorities) and the primary outcome is associated with those populations.
There is a risk of attribution or stigmatization toward First Nations, Métis or Inuit individuals or communities
This includes situations where there is an over representation of First Nations, Métis, or Inuit individuals in the study cohort or population. MCHP understands that a risk for attribution may not be evident when starting out on a research project; if this changes during the project then input and advice will be sought before continuing with the research.
The MCHP Repository Access Unit (RAU) will assist you with the contact information for requesting approvals / permissions from First Nation, Métis and/or Inuit representatives. Partnerships should be identified and included before submitting for funding or seeking approvals.
5. Project Specific Data Approvals (if applicable)
If your study uses project specific data, it is the Principal Investigator's (PI's) responsibility to get written approval from the data provider to use the data.
For more information, review the Guidelines for requests for project specific data (PDF)
Private Sector / Industry-Sponsored Research Projects
For private sector / industry-sponsored research projects using the Repository, MCHP will assist in facilitating approvals from PHRPC, HREB, non-MH data providers, First Nations, Métis, and/or Inuit representatives.
Tasks
The required tasks include:
- Complete the PHRPC Application form
- Complete the UM HREB Application form
- Complete non-MH Data Provider approval forms (if applicable). MCHP will provide you with the current templates for obtaining approvals from each of the non-MH data providers. Please complete the templates and return them to MCHP who will facilitate the submission of these forms and any required supporting documentation to the appropriate organizations.
- For project specific data, the Data Sharing Agreement or letter of approval is sufficient (if applicable).
- Forward copies of ALL the completed documentation to MCHP This includes: approval/permission application forms, subsequent approvals, and any additional supporting documentation.
- MCHP will Submit Documentation for Final Approval from PHRPC - once all the required approval/permission documentation is received at MCHP from all organizations involved, this documentation will be submitted to PHRPC, by MCHP, for review and to obtain the final PHRPC approval for the research project.
- PHRPC Final Approval Notification - PHRPC will notify the researcher and MCHP when they provide final approval for the research project.
Additional tasks for students
Graduate students must have their research project approved by their committee prior to submitting any other approvals. For more information, review the Guidelines for student use of the repository (PDF)
Contact
For more information, contact the MCHP Repository Access Unit.
Step 6: Researcher agreement and approval documentation submission
Overview
The researcher agreement is a document that outlines the obligations and responsibilities of all parties with respect to accessing and performing research with the Manitoba Population Research Data Repository.
The parties involved in the researcher agreement are:
- the Manitoba Government (Manitoba Health)
- the University of Manitoba (Manitoba Centre for Health Policy)
- the researcher (principal investigator)/research project team
Researcher agreements are approved for a time period of four years, with a possible two year extension.
About the agreement
It is important to note that the most current template of the agreement must be used, or your proposal may be delayed or outright rejected.
The Manitoba Centre for Health Policy (MCHP) will provide you with the researcher agreement, or student researcher agreement, along with guidelines for completion.
The researcher agreement must be signed by the principal investigator/student and submitted to MCHP after you have received all the necessary approval/permission documentation for your research project.
Approval documentation is the "confirmation of approval/permission letter/documentation" you receive from:
- Provincial Health Research Privacy Committee (PHRPC)
- University of Manitoba (UM) Health Research Ethics Board (HREB)
- non-Manitoba Health data providers (if applicable)
- First Nations, Métis or Inuit representatives (if applicable)
- the project specific data provider, if the data is not part of the Manitoba Population Research Data Repository (if applicable)
MCHP will facilitate the completion of the researcher agreement/student researcher agreement after you have received the final PHRPC approval notice.
You must forward all the required project approval documentation, including the completed researcher agreement/student researcher agreement, to the MCHP Repository Access Unit (RAU).
Tasks
The required tasks include:
1. MCHP to provide the researcher agreement/student researcher agreement form
Once the Provincial Health Research Privacy Committee (PHRPC) notifies the principal investigator (PI) of final approval for the research project, please contact the Manitoba Centre for Health Policy (MCHP) Repository Access Unit (RAU). MCHP will then provide you with the MCHP researcher agreement, or student researcher agreement, along with guidelines for completion. The most current template of the agreement must be used, or your proposal may be delayed or outright rejected.
2. Complete the researcher agreement/student researcher agreement and submit all relevant project approvals to MCHP
Please send an electronic copy, or two signed, paper (hard) copies, of all the following documentation to the MCHP Repository Access Unit (RAU):
- the completed researcher agreement/student researcher agreement
- the completed Provincial Health Research Privacy Committee (PHRPC) submission, including a copy of the data table and proof of funding
- the University of Manitoba (UM) Health Research Ethics Board (HREB) letter of approval to proceed with study
- any non-Manitoba Health data provider approvals to proceed with the study (if applicable)
- any First Nations, Metis or Inuit approval letters/permissions to proceed with the study (if applicable)
- any additional documentation (e.g., data sharing agreement, letter of approval) that provides permission for using project specific data that is not part of the Manitoba Population Research Data Repository (if applicable)
- the PHRPC conditional approval letter, your response to the conditions (including any attachments that were submitted), and the final PHRPC letter of approval to proceed with the study
Additional tasks for students
Submit the approval letter from your thesis committee.
Notes:
- Please allow three weeks for processing
- Copies of all submissions to all organizations, and the subsequent approvals, must be forwarded to MCHP
- All submissions must identify the same principal investigator and project title, and include the same data request table. Failure to do so may result in a delay to the start of your research project.
Contact
For more information, contact the MCHP Repository Access Unit (RAU).
Step 7: Project initiation and analysis plan development
Overview
Once all of the approvals and a completed researcher agreement are received at the Manitoba Centre for Health Policy (MCHP), they will begin the project initiation process.
About the initiation process
The initiation process includes the following tasks below.
1. Contacting the principal investigator/research team to confirm the data and analytic requirements before starting any processing. If necessary, a meeting will be arranged.
2. Manitoba Centre for Health Policy (MCHP) suggests developing a project analysis plan to facilitate a clear vision for the project for all team members. For more information, please see the following documents:
- Guide - MCHP Analysis plan process (PDF)
- Example - MCHP Analysis Plan (PDF)
- Template - MCHP Analysis Plan (Word)
3. Depending on the arrangements for the project, MCHP will:
- set up an accreditation session, if necessary
- allocate network/system access, space (virtual and physical) and setting up access as required from the proposal.
- identify the MCHP analyst who will be working with the project team and communicate this information to the principal investigator (if required)
- if a member of the project research team will be doing the analysis, this requires access to the MCHP secure network/analytic systems. An account will need to be set up and configured by completing the MCHP computer application/access form and sending it to the MCHP Repository Access Unit so the account can be set up. NOTE: There is a system access fee associated with each user account.
- extract the required data from the Data Repository and make it available to the project team in a secured location.
- if the project is using project specific data (non-Repository data), once the linkage/crosswalk data is received from Manitoba Health (MH) and the service data is received from the data provider, it will be uploaded to the MCHP secure network and made available to the project team.
- complete the required analysis (if required).
4. The MCHP finance team will contact the principal investigator to discuss finances and determine the billing/payment arrangements for the project.
Tasks
A meeting will be arranged with the Manitoba Centre for Health Policy (MCHP) to discuss and confirm the data and analytical requirements for your research project, including the development of a project analysis plan.
MCHP will:
- allocate network/system access, confirm accommodation/space requirements, and setup computer/access as required.
- extract and setup access to the Repository data, as required.
If the project is using project specific data (non-Repository data), MCHP will upload it when it arrives from Manitoba Health and setup access to it.
If required, MCHP will complete the required analysis for the project.
Finances
The following information describes the financial requirements and the billing/payment process used by the Manitoba Centre for Health Policy (MCHP) for your research project.
A member of the MCHP finance team will contact the principal investigator to confirm the project billing information (e.g., University of Manitoba (UM) FOAP (Fund Organization Account Program) account or payment information for externally funded projects).
Some projects have a set system access fee for use and access to the MCHP computer/analytic systems. This includes projects that have remote access or involve private sector/industry sponsored funding.
This system access fee is invoiced once per year and is based on a system use agreement. This annual fee covers the back-end support required to maintain the Data Repository, computer systems, and network security. Sites or users without a predefined access fee are invoiced a system access fee for each project. Students are exempt from paying the system access fee.
Work that is completed by MCHP staff will be invoiced once every month for the hours they spend on the project during the prior month. The hourly rate is the current rate as identified in the current accreditation material. There is a student rate for student-led projects.
NOTE
The number of hours estimated, and project hours used, can be queried from either the analyst working on the project or Charles Burchill at any time.
If an invoice remains outstanding for more than a month, a reminder will be sent to the principal investigator. If the invoice is not paid after two months, the project will be put on hold and MCHP will not perform any further work on the project.
If three months or more has passed and the invoice is still not paid, the project will be archived and an invoice will be sent to the Dean or Unit Director of the principal investigator.
If the archived project is the only project associated with a researcher or analyst on our system then the userid will be locked.
Projects may continue to have active approvals so the researcher can continue to write and publish manuscripts or complete presentations, but access to MCHP systems and analysis will be restricted.
Contact
For more information, contact the MCHP Repository Access Unit.
You may also contact:
Step 8: Amendments
Overview
An amendment is a significant change in your original, approved research project that must go through a review and approval process. Please submit amendments after all aspects of the original proposal and initial final approval process have been completed.
NOTE
Amendments to your proposal and the original approvals will not be accepted by MCHP during the initial proposal and approval process. Submitting an amendment prior to final approval being in place may lead to delays in project commencement.
About the amendment process
If any of the following project information changes, you must submit an amendment and await approval before you can proceed with the changes to your research project:
- the years of data for your study
- datasets/data sources
- the research question(s)/objective(s)
- the location of your data storage and/or analysis
- the funding source or sponsor
- the investigators or team members involved in your research project
NOTE
The Provincial Health Research Privacy Committee (PHRPC) requires notification of any changes in the original submission with respect to investigators or anyone accessing line-level data. Notification is also required if someone other than one of the investigators becomes first author on a manuscript, regardless of whether the individual has access to line-level data. If you have questions, please check with the Research Manitoba RITHIM Coordinator.
If an amendment to your project is required, please contact the Repository Access Unit (RAU) with a short description of the changes you are making. They will guide you through the amendment process.
An amendment request will need to be sent to each of the following groups for review and approval:
Provincial Health Research Privacy Committee (PHRPC)
- Please see the links in the Forms and Guides section on the PHRPC Submission Information web page for access to:
- the PHRPC protocol amendment form, and
- Guidelines for submitting a HIPC/PHRPC protocol amendment
University of Manitoba (UM) Health Research Ethics Board (HREB)
- Please see the HREB Submission requirements web page for general submission information.
- For specific amendment requirement information, please read the following:
All non-Manitoba Health (non-MH) data providers involved in your project (if applicable)
The RAU will provide you with a current template for submitting an amendment to non-MH data providers. Please complete the template and return it to the RAU, who will facilitate the amendment submission.
First Nations, Métis and Inuit representatives (if applicable).
The RAU will provide contact information for information on the current process to complete an amendment. For more information, please visit First Nations Health and Social Secretariat of Manitoba - Health Information Research Governance Committee (HIRGC).
Project specific data providers (if applicable).
The RAU will provide you with a current template for submitting an amendment to project specific data providers. Please complete the template and return it to the RAU, who will facilitate the amendment submission.
Tasks
The required tasks include:
- Contact the Repository Access Unit (RAU) with the details regarding your amendment
- Submit a Provincial Health Research Privacy Committee (PHRPC) protocol amendment form to the PHRPC - see the Forms and Guides section on the PHRPC Submission Information web page for more information
- Submit the appropriate forms to the University of Manitoba Bannatyne Campus Health Research Ethics Board (HREB) - see the section titled Protocol amendments or changes to study for more information
- Complete the template letters outlining the changes to the non-Manitoba Health data providers (if applicable). The RAU will submit these letters on your behalf
- Submit a letter outlining the changes to the project specific data provider (if applicable)
- Send a copy of all amendment approval documentation from PHRPC, HREB, and all other data providers involved to the RAU
Contact
For more information, contact
Step 9: Project reporting and annual renewals
Overview
The University of Manitoba (UM) Health Research Ethics Board (HREB) requires an annual study status report to be submitted.
An annual MCHP Accreditation renewal is also required. To ensure your project is able to continue, you will receive a reminder each year via email.
Please attend to these annual tasks promptly. If you do not submit an Annual Report or renew your accreditation, there is a risk of your project being closed.
At any time during your project, if a project team member with access to the MCHP systems leaves, or moves to a different position, please notify the MCHP Repository Access Unit.
Tasks
The required tasks include:
- Complete an annual study status report
- Submit the annual study status report to University of Manitoba (UM) Health Research Ethics Board (HREB)
- Send a copy of the HREB certificate of annual approval to the Provincial Health Research Privacy Committee (PHRPC)
- Send a copy of the HREB certificate of annual approval to the MCHP Repository Access Unit (RAU)
- Complete the annual MCHP accreditation renewal - see Step 1: Attend accreditation session for more information
- Notify MCHP if a team member with access to the MCHP systems leaves, or moves to a different position
The following information can be found on the University of Manitoba (UM) Office of Research Ethics and Compliance - Annual review procedures web page, including:
- a description of the annual review process
- a list of Required HREB forms, including the Annual Study Status Report Form
- a description of the process for submitting your Annual Study Status Report (ASSR)
Contact
For more information, contact the MCHP Repository Access Unit.
Step 10: Data use and output guidelines
Overview
All studies using data from the Manitoba Population Research Data Repository must follow the guidelines and requirements described in this section.
The principal investigator/student who has signed a researcher agreement/student researcher agreement is responsible for ensuring the obligations outlined in the approvals and agreements are followed by all project team members.
NOTE: There are specific guidelines and requirements for output (e.g., publications and presentations) related to research projects using data from the Repository, as requested from our data providers and approving agencies.
Terminology and definitions
"Use" - refers to the development of analytic results and creation of temporary data analytic files related to data approved for access for the study.
Use is limited to:
- the approved study objectives
- those study members with the appropriate approvals and permission
"Disclosure"/"Release" - refers to the transfer of results or information that has been generated using data from the MCHP Data Repository out of the secure MCHP environment. This includes release of information or output for public dissemination, in either hard copy or electronic form.
Dissemination of such information is governed by organizations such as the following:
- Government legislation:
- health data - information sharing and protection of Privacy Agreement under the Personal Health Information Act (PHIA)
- non-health data such as Manitoba Families and Manitoba Education data under the Freedom of Information and Protection of Privacy Act (FIPPA)
- University of Manitoba / Manitoba Centre for Health Policy policies and procedures.
- Data providers for the data used in the study producing the output (e.g., Manitoba Health data, Department of Justice data).
"Output" - refers to material generated from the Data Repository subject to rules of disclosure. This material includes:
- manuscripts for publication (including thesis publications) or other printed media
- posters and abstracts
- oral presentations
- printed materials distributed at educational or professional conferences or seminars
- website postings of project related material
Note: Where output has been approved by Manitoba Health and other providers, its inclusion in multiple presentations does not require additional approvals for each subsequent presentation. However, Manitoba Health and other data providers should still be notified regarding each presentation.
When multiple presentations of content are likely to occur, it is suggested that a deck of slides be compiled for initial review by Manitoba Health to avoid the need for submitting content for approval for every individual presentation.
Guidelines and requirements
All output prepared from data in the Manitoba Centre for Health Policy (MCHP) Data Repository for public release must adhere to the following guidelines:
- Submission of output notification and approval process
- Suppression of small data values
- Inclusion of required acknowledgements and disclaimers
- Submission of data underlying findings in manuscripts to a public repository
- Authorship guidelines
Note: this does not apply to material presented or circulated internally to authorized project team members.
1. Submission of output notification and approval process
For Manitoba Health (MH) data, output for public release produced using MH data must be handled as described below.
Thirty days prior to release for publications "in learned journals or thesis presentation" and 30 days prior to release for conference/meeting presentations and for abstracts, the output must be sent to Manitoba Health and MCHP:
1. Manitoba Health for review regarding confidentiality, privacy and consistency with the Provincial Health Research Privacy Committee’s (PHRPC's) approved protocol.
The letter (or email) should include the following clause:
- Enclosed is a manuscript, which has recently been completed, titled [title of manuscript]. I would appreciate it if you would review this paper with regard to the confidentiality of personal health information. I would also welcome any other critique that you might have in regards to the use of the health data in this paper.
Following publication, the principal investigator must send a copy of the publication to MH in PDF form. If the dissemination of output will be entirely verbal (no publication of abstract or proceedings), no formal submission for approval to MH is required.
2. MCHP - attention of the MCHP Repository Access Unit (RAU)
- In addition, if there is a planned media release, please inform info@cpe.umanitoba.ca of the media release details.
For non-Manitoba Health (non-MH) data
Output produced using non-MH data has the following requirements:
- 30 days prior to public release, the data provider(s) must be notified of the planned release. MCHP will provide you with the contact information for the non-MH data providers.
- Following publication, the principal investigator must send a copy of the publication / presentation to the data provider.
- Prior to public release, a letter of acknowledgement/approval must first be obtained from PHRPC and non-MH data providers. If pre-released materials are approved for limited distribution they should be clearly stamped PRELIMINARY - NOT FOR FURTHER DISTRIBUTION. They should not be stamped CONFIDENTIAL.
For private/industry sponsored research projects
The following output requirements relate to private/industry sponsored research projects:
- The principal investigator will provide the sponsor with a final research report, if outlined in the funding contract/proposal, 60 days prior to public release. No preliminary outcomes or information will be provided to the sponsor or their representative prior to the final research report being available.
- The principal investigator will provide all of the data providers an opportunity for a verbal presentation regarding the project findings and conclusions, and content of all reports or manuscripts, at least 60 days in advance of the public release of such reports or manuscripts.
- The sponsor will not have the opportunity to influence the data analysis or interpretation, study conclusion or content of any reports or manuscripts.
2. Suppression of small data values
- All output for public release must ensure that the data are aggregate, and summarized in such a way that any cell or record value represents at least six or more individuals or events. A value of zero is also acceptable.
- When using the Immigration Permanent Resident Database (IRCC) data, please ensure that the data are aggregate, and summarized in such a way that any cell or record value represents at least 10 or more individuals or events. A value of zero is also acceptable.
- Additionally, it must be ensured that the suppressed information cannot be obtained by other calculations (e.g., totals from which the individual cell values can be inferred).
- The following information is available from MCHP upon request:
- Policy - suppression of small data values
- Examples and illustrations - How to handle small sample size and cell suppression
- Suppression strategies - April 14, 2010 programmers meeting notes
3. Acknowledgements and disclaimers
There are a number of acknowledgements and disclaimers that should be reviewed to see if they need to be included in your publication / presentation. This includes information on copyright, referencing SAS, use of adjusted clinical groups (ACGs), use of Canada census data, and a statement on Traditional Territories. For more detailed information, please read both the Acknowledgments and Disclaimers sections on our website.
The following standard statement must be used in all publications:
The authors acknowledge the Manitoba Centre for Health Policy for use of data contained in the Manitoba Population Research Data Repository under project #_____ (HIPC#____ or PHRPC#____). The results and conclusions are those of the authors and no official endorsement by the Manitoba Centre for Health Policy, Manitoba Health, or other data providers is intended or should be inferred. Data used in this study are from the Manitoba Population Research Data Repository housed at the Manitoba Centre for Health Policy, University of Manitoba and were derived from data provided by Manitoba Health and (names of other data providers).
NOTE: The names of data providers can be found in the Manitoba Population Research Data Repository Data List.
The statement above should be substituted with the following statement in all publications that have received private sector/industry sponsorship:
We acknowledge the financial support of (name of sponsor). The authors acknowledge the Manitoba Centre for Health Policy for use of data contained in the Manitoba Population Research Data Repository under project #_____ (HIPC#_____ or PHRPC#_____). The results and conclusions are those of the authors and no official endorsement by the Manitoba Centre for Health Policy, Manitoba Health, University of Manitoba, other data providers, and the (name of sponsor) is intended or should be inferred. Data used in this study are from the Manitoba Population Research Data Repository housed at the Manitoba Centre for Health Policy, University of Manitoba and were derived from data provided by Manitoba Health and (names of other providers).
4. Submission of data underlying findings in manuscripts to a public repository
Many journals now require that source data used in analysis for articles be submitted to a publicly accessible repository, such as PLOS Journals. This is not possible for most analysis done at MCHP as disclosure of any portion of the MCHP Repository for use/access outside of MCHP is not permitted.
An example of a data availability statement (see below), provides a response to journals requesting source data from the MCHP Repository.
Journals may still require submission of statistical or aggregate data not in the publication; they will provide recommended sites and processes for submission.
The University of Manitoba's Dataverse is a suitable open source repository for the management and dissemination of research data. Please contact the University of Manitoba Libraries Research Services Team for more information.
Example of data availability statement
Data used in this article was derived from administrative health and social data as a secondary use. The data was provided under specific data sharing agreements only for approved use at Manitoba Centre for Health Policy (MCHP). The original source data is not owned by the researchers or MCHP and as such cannot be provided to a public repository. The original data source and approval for use has been noted in the acknowledgments of the article. Where necessary, source data specific to this article or project may be reviewed at MCHP with the consent of the original data providers, along with the required privacy and ethical review bodies. Statistical and anonymous aggregate data associated with this paper, along with metadata describing the original source, has been submitted to (name of site repository).
5. MCHP authorship guidelines
MCHP has developed Guidelines for authorship (PDF) for publications.
In addition, the University of Manitoba has a policy titled Responsible Conduct of Research - Code of Ethics (PDF) that mentions guidelines around authorship in section 2.4.
Tasks
The required tasks include:
Output notification - for Manitoba Health (MH) and Manitoba Centre for Health Policy (MCHP) - provide MH and MCHP with notice, within the appropriate time frame (e.g., 30 days prior to), of your plan to publish or present output from your study.
Contact information:
- Manitoba Health - Provincial Information Management and Analytics (PIMA) - pima@gov.mb.ca
- MCHP Repository Access Unit - mchp_access@cpe.umanitoba.ca
Output notification - for non-MH data providers - provide non-MH data providers with notice, within the appropriate time frame (e.g., 30 days prior to), of your plan to publish or present output from your study. MCHP will facilitate this process.
Output approval
MH and non-MH data providers will provide a letter of acknowledgement/approval for the release of output from your research project.
Media release notification (if applicable)
Please inform info@cpe.umanitoba.ca of upcoming media releases related to your research project.
Send copy of output to MH and non-MH data providers
Following publication, send a copy of the publication to MH and the non-MH data providers;
Adhere to the general data use and output guidelines
The guidelines are outlined on this web page, and more specifically to:
- Suppress data values less than six; zero values are acceptable (applies to all Repository data, except the following)
- When using the Immigration Permanent Resident Database (IRCC) data, suppress data values less than 10; zero values are acceptable
- Use appropriate acknowledgements and disclaimers
Use of data availability statement
If required, providing a "data availability statement" to journals now requesting source data from your project. You may still be required to submit statistical or aggregate data that is not in the publication.
Contact
For more information, contact the MCHP Repository Access Unit.
Step 11: Project closure
Overview
Project closure signifies that the data analysis component of the research project is complete and the principal investigator/data analyst will not require further access to the data again.
A project is officially closed when the principal investigator notifies the MCHP Repository Access Unit that the project is complete.
Tasks
Upon closure, the principal investigator must complete a Final Study Status Report (FSSR) Form and send it to:
- University of Manitoba (UM) Health Research Ethics Board (HREB)
- Manitoba Centre for Health Policy (MCHP) Repository Access Unit
See the University of Manitoba (UM) Health Research Ethics Board (HREB) section titled Study closure and termination for more information and access to the Final Study Status Report (FSSR) Form.
Projects may continue to have active approvals so the researcher can continue to write and publish manuscripts or complete presentations, but access to the data on MCHP systems and any analysis will be restricted. If a team member with access to the MCHP systems leaves, or moves to a different position, please notify MCHP immediately.
Your research project may be closed, if:
- you do not complete the MCHP annual accreditation renewal
- the annual HREB reporting requirements have not been satisfied and/or such reports have not been sent to the MCHP Repository Access Unit (RAU)
- the invoiced amount has not been paid for two months.
Your research project may be archived, if:
- the invoiced amount has not been paid and three months or more have passed
- When a project is archived, it is removed from the MCHP system and stored for seven years. MCHP requires renewed or updated approvals from the Provincial Health Research Privacy Committee (PHRPC), HREB and all data providers in order to restore data for the project
Contact
For more information, contact the MCHP Repository Access Unit.
Guidelines, forms & documents
Guidelines
Concept dictionary and glossary
Browse/Search the Concept Dictionary and Glossary
The Concept Dictionary and Glossary represent a series of tools that have been developed at MCHP to permit application of a variety of theoretical frameworks, analytical techniques, statistical approaches and mapping resources appropriate for use with administrative databases.
Concept Dictionary - describes over 300 research concepts developed at MCHP for analyzing data contained in the data repository. These detailed operational definitions of variables or measures include a description of the data, approach and methods used, programming tips/cautions, SAS code (where not restricted for internal use), discussion of known issues, additional readings, and reference information.
Glossary - documents terms commonly used in population based research. Each term contains a brief definition, links to related terms and concepts in the dictionary, reference information, as well as links to external sites.
Historic language use
Please note that terms in the Manitoba Centre for Health Policy’s Concept Dictionary and Glossary for Population Based Research span over 30 years of research and therefore some older studies and concept descriptions may use outdated or insensitive language. These words reflect the culture and language at the time of publication and do not reflect the centre's current values and beliefs.
Alternative entry points
- Health Measures Concept Search
- Educational Measurement Concept Search
- Geographical Analysis Concept Search
Study design and methods
A general study planning tool, with information on study design, eligibility criteria, data sources, study period, theoretical frameworks, and links to methodological and statistical tools.
Additional information
- Current concepts in progress (PDF) - an advance look at upcoming releases of new work or concepts to be modified - last updated January 12, 2024.
- Completed concepts (PDF) - a list of concept work completed since December 21, 2022 - last updated January 12, 2024.
Concept Dictionary contributors
This resource was created to assist users of the extensive, rich, Data Repository housed at MCHP. The Concept Dictionary arose at MCHP out of a desire to help researchers carry out methodologically sound research using consistent, validated algorithms. In turn, researchers who share their discoveries help to grow and improve the value of this publicly-accessible resource.
We are grateful to all researchers and analysts who contribute their time and effort to this resource by creating new concepts and/or updating existing concepts. We encourage contributions to the Concept Dictionary; guidelines for concept development are described in the Concept development template (PDF).
For more information, email info@cpe.umanitoba.ca.
Data quality
For each new Repository data acquisition, and any updates to existing Repository data, MCHP performs a data quality evaluation process on the data and produces a data quality report.
Please review the following documentation to become more familiar with the MCHP data quality evaluation process:
MCHP's data quality process is based on work developed and reported in the 2012 MCHP deliverable "A systematic investigation of Manitoba's provincial laboratory data" (PDF).
MCHP has developed macros that automate the data quality evaluation process. These macros are available for development, use, and distribution under a GNU General Public License (version 3.0) (TXT). All macros are written in the SAS® programming language with the exception of the AUTOMATE macro (an Excel VBA macro).
Pre data quality macros
These two macros should be run prior to running any of the Main or Intermediate data quality macros.
1. PREDQ Macro (TXT)
This macro produces a summary report containing the number of non-missing records in each variable for two different versions of the same dataset. For more descriptive information, see the accompanying documentation for the PREDQ SAS Macro directly below.
2. META Macro (TXT)
This macro generates a metadata data set that contains information about variables including their associated formats. Once generated, this data set can be used in data quality and documentation processes.
Main data quality macros
There are several macros, including:
CONTENTS Macro (TXT)
This macro generates an overview table that contains dataset names, dataset labels, and the number of records and fields for a series of datasets.
VIMO Macro (TXT)
This macro generates a VIMO (valid, invalid, missing, outlier) table in Microsoft Excel format for a specific dataset.
LINK Macro (TXT)
This macro creates a table that shows the number and percentage of linkable records of a specific dataset or list of datasets. If the dataset is a cluster dataset, this macro can be run for a list of cluster members. This macro also generates a distribution table for the PHINTYPE variable.
LINKYR Macro (TXT)
This macro displays the percentage of linkable records over fiscal/calendar year for a specific dataset or a list of datasets.
AGREEMENT Macro (TXT)
This macro checks the agreement between a list of datasets and registry data for the same individuals and produces Kappa Statistics for sex and date of birth.
TREND Macro (TXT)
This macro performs a trend analysis over a specified time range, the output is saved in graphic format (.PNG)
REFERENTIAL INTEGRITY CHECK Macro (TXT)
This macro performs a referential integrity check between two databases. Duplicate or missing primary key and foreign key values found in the foreign table but not in the primary table are identified and the result is summarized in RTF format.
VALIDATION Macro (TXT)
This macro scans the dataset and counts the number of data inconsistencies based on the validation rules specified by the user. The result is output in RTF format.
Intermediate data quality macros
INVALID CHECK Macro (TXT)
This intermediate macro compares values of variables against values in their associated format within a specific dataset. Any values not in their associated format are identified and output to the VIMO table as invalid values. This macro is called from within the DQ_VIMO macro.
Post data quality macros
GEN Macro (TXT)
This post data quality macro has been developed as a bridge between SAS and EXCEL so that the user can automatically generate a well-formatted excel output from within SAS. This macro requires the automate.bas Excel VBA macro in order for it to work.
Auxiliary excel VBA macro
AUTOMATE Macro (TXT)
This Excel VBA Macro takes in plain and unformatted Excel files produced by DQ_VIMO, DQ_LINK, DQ_LINKYR and DQ_AGREEMENT and produces well-formatted EXCEL outputs that are used to generate the data quality report. This Excel VBA Macro is used by the DQ_GEN macro.
Getting started
You will require a PC file server.
Using 'Save Target As,' download all macros including dq_automate.bas into the same folder.
Remove the .txt file extensions from each SAS macro.
Note: dq_agreement, dq_link and dq_linkyr macros are written to work for MCHP data only. It will not work for non MCHP data.
More about the Data Repository
Study design and methods
The databases housed at MCHP permit a large range of population-based observational research. At the same time they are complex and can present unique methodological challenges. The following information, including general study planning considerations, can help researchers deal with these challenges.
Types of studies
Cross-sectional, case-control, and cohort studies are the most commonly used observational designs for studies using data from the MCHP Data Repository. Multiple outcome variables can be examined and data collection tends to be relatively inexpensive. At the same time, issues can include possible loss to follow-up as well as longitudinal changes in coding, databases, and data fields.
Study designs can be descriptive or analytical:
- Descriptive studies measure disease occurrence, health service use, or risk factor prevalence in populations, seeking to identify personal characteristics, geographic locations, and time periods associated with an unusually high or low risk of events. Population-based descriptive studies provide the most accurate quantitative estimates of disease frequency/exposure prevalence or health service use in various populations. (Ecological studies are a type of descriptive study using aggregate statistics on populations. A fundamental limitation of ecological studies is that they consider only aggregate data for an entire population and not whether the disease and exposure of interest occurred in the same person.)
- Analytical studies measure the association between disease or service use and exposure in individuals or populations. identifying the specific factors that cause increased or decreased risk.
While cross-sectional studies collect information for a single point in time, case control and cohort studies can be either retrospective or prospective. The former poses a question and looks back whereas the latter poses a question and looks forwards (Institute for Work and Health, 2010):
A retrospective study "follows up" to view outcomes using data that has already been collected (frequently for other purposes) from past records or from a person's recollections. Issues for this type of study include recall bias and limits to the amount of data that can be collected on one occasion. (Mann 2003).
A prospective study follows individuals forward through time to see if they develop the outcome of interest. (Mann 2003). Intra-subject variability tends to be smaller than inter-subject variability for this type of study, which also allows for separation of aging/maturation effects from cohort effects.
References
- CJ Mann. Observational research methods. Research design II: cohort, cross sectional, and case-control studies. Emerg Medical Journal, 2003 Jan;20(1):54-60. doi: 10.1136/emj.20.1.54.
- Institute for Work and Health. What researchers mean by ... retrospective vs prospective studies. Atwork newsletter. Issue 59, Winter 2010. [accessed July 6, 2011].
Theoretical frameworks
Theoretical frameworks help define the scope of research; they should "facilitate the presentation of evidence in such a way as to make its implications more apparent" (Evans, 1990). The following are examples of frameworks that have previously been used in MCHP studies:
A. Health Services Utilization Framework - Andersen and Newman
The purpose of this framework is to discover conditions that either facilitate or impede utilization. The goal being, to develop a behavioural model that provides measures of access to medical care. The framework was first developed in the 1960s and has since gone through four phases. Developed in the 1990s, the framework below represents the fourth phase.
An individual's access to and use of health services is considered to be a function of three characteristics:
1) Predisposing Factors: The socio-cultural characteristics of individuals that exist prior to their illness.
- Social Structure: Education, occupation, ethnicity, social networks, social interactions, and culture
- Health Beliefs: Attitudes, values, and knowledge that people have concerning and towards the health care system
- Demographic: Age and Gender
2) Enabling Factors: The logistical aspects of obtaining care.
- Personal/Family: The means and know how to access health services, income, health insurance, a regular source of care, travel, extent and quality of social relationships
- Community: Available health personnel and facilities, and waiting time
- Possible additions: Genetic factors and psychological characteristics
3) Need Factors: The most immediate cause of health service use, from functional and health problems that generate the need for health care services. "Perceived need will better help to understand care-seeking and adherence to a medical regimen, while evaluated need will be more closely related to the kind and amount of treatment that will be provided after a patient has presented to a medical care provider." (Andersen, 1995)
- Perceived: "How people view their own general health and functional state, as well as how they experience symptoms of illness, pain, and worries about their health and whether or not they judge their problems to be of sufficient importance and magnitude to seek professional help." (Andersen, 1995)
- Evaluated: "Represents professional judgment about people's health status and their need for medical care." (Andersen, 1995)
References
- Aday LA, Andersen RM. A framework for the study of access to medical care. Health Serv Res 1974;9(3):208-220.
- Aday LA, Andersen RM. Equity to access to medical care: a conceptual and empirical overview. Med Care 1981;19(supplement):4-27.
- Andersen RM, Newman JF. Societal and individual determinants of medical care utilization in the United States. Milbank Memorial Fund Quarterly– Health and Society 1973;51(1):95-124.
- Andersen RM. Revisiting the behavioral model and access to medical care: does it matter? J Health Soc Behav 1995;36(March):1-10. [Abstract - Revisiting the behavioral model and access to medical care: does it matter?]
MCHP Studies Using Framework:
- Lix L, Finlayson G, Yogendran M, Bond R, Bodnarchuk J & Soodeen R. "Primary Prevention: An Examination of Data Capabilities in Manitoba." January, 2005. [Full Report - Primary Prevention: An Examination of Data Capabilities in Manitoba.] [Summary - An Apple a Day: Primary Prevention Data in Manitoba]
- Tomiak M, Berthelot J, Guimond E & Mustard C. Factors associated with nursing-home entry for elders in Manitoba, Canada. Journal of Gerontology: Medical Sciences 2000; 55A(5): M279-M287. [Abstract - Factors associated with nursing-home entry for elders in Manitoba, Canada]
B. Determinants of Health Framework - Evans and Stoddart
An intellectual framework of the interaction between determinants of health, with the goal of understanding the health of a population beyond the bounds of the health care system. Evans et al. (1990) sought to "construct an analytic framework within which such evidence can be fitted, and which will highlight the ways in which different types of factors and forces can interact to bear on different conceptualizations of health."
References
- Evans RG, Stoddart GL. Producing health, consuming health care. Soc Sci Med 1990;31(12):1347-63.
- Evans RG, Stoddart GL. Consuming research, producing policy? Am J Public Health. 2003 Mar;93(3):371-9.
C. Pan Canadian Healthy Living Strategy Framework
In September 2002, the Federal/Provincial/Territorial (F/P/T) Ministers of Health announced that they agreed to work together on an Integrated Pan-Canadian Healthy Living Strategy. The Healthy Living Strategy is an initiative aimed at reducing non-communicable diseases by addressing their common risk factors and the underlying conditions in society that contribute to them.
"This study provides a strategic foundation for expanding and enhancing existing policies and programs, building new linkages and moving forward in a strategic way to advance healthy living goals and objectives." (Minister of Health, 2005) The goals of the strategy were to improve overall health outcomes and reduce health disparities.
References
- The Canadian Minister of Health. "The Integrated Pan-Canadian Healthy Living Strategy Framework." (2005)
MCHP Studies Using Framework:
- Lix L, Finlayson G, Yogendran M, Bond R, Bodnarchuk J & Soodeen R. "Primary Prevention: An Examination of Data Capabilities in Manitoba." January, 2005. [Full Report - Primary Prevention: An Examination of Data Capabilities in Manitoba.] [Summary - An Apple a Day: Primary Prevention Data in Manitoba]
D. Canadian Health Indicators Framework
Developed from a national consensus conference in 1999, these Canadian Health Indicators are used mainly by the Canadian Institute for Health Information, in their 2002-2005 Health Indicators Reports, and by Statistics Canada to measure health and the health system in Canada.
Statistics Canada looked at several health indicators, including:
- Health status - How healthy are Canadians? Health status can be measured in a variety of ways, including well-being, health conditions, disability, or death.
- Non-medical determinants of health - Non-medical determinants of health are known to affect our health and, in some cases, when and how we use health care.
- Health system performance - How healthy is the health care system? These indicators measure various aspects of the quality of health care.
- Community and health system characteristics - These measures provide useful contextual information, but are not direct measures of health status or the quality of health care.
References
- Canadian Institute for Health Information: Health indicators 2005 Ottawa: CIHI; 2004.
- Canadian Institute for Health Information: Health indicators 2005: Definitions, data sources, and rationale Ottawa: CIHI; 2004.
- Canadian Institute for Health Information and Statistics Canada. Health indicators 2004, no.1 Ottawa: CIHI; 2004
MCHP Studies Using Framework
- Lix L, Finlayson G, Yogendran M, Bond R, Bodnarchuk J & Soodeen R. "Primary Prevention: An Examination of Data Capabilities in Manitoba." January, 2005. [Full Report - Primary Prevention: An Examination of Data Capabilities in Manitoba.] [Summary - An Apple a Day: Primary Prevention Data in Manitoba]
Study design/methodology resources
Study design descriptions advantages and disadvantages - see Types of Study Design
Defining the study sample/population [see Other considerations] - includes information on:
- What are the demographics of the population/sample to be studied? Age, sex and residence might be specified, for example, as men aged 65+ resident in Manitoba.
- What are the treatment characteristics (disease/procedure, provider)? This might be specified, for example, as hospital admissions in Winnipeg for acute myocardial infarction defined as ICD-9-CM 410.
- Will small area analysis be needed for location of residence and/or for provider?
Other questions include:
- What is the unit of analysis? Will a cohort need to be developed?
- How will issues characteristic of the study design (e.g., loss to follow-up for longitudinal studies) be dealt with?
- Has work been done previously using the study design on MCHP data?
- Longitudinal design issues - Changes in data values occurring over time need to be taken into account in longitudinal study design. They include loss to follow-up (e.g., residential moves, death), classification coding changes, family structure alterations, etc.
Concepts have been developed to assist the researcher with information on handling these issues:
Classification schemes
Multiple codes within various time frames (as opposed to a code at one point in time) will require extra programming. See the following concepts for more information: ICD Coding in MCHP Data and ICD-10 to ICD-9-CM conversion
Statistical tools/issues
- Statistics for Large Databases - general statistical tools and approaches for working with regression analysis, GEE (generalized estimating equations), sensitivity and specificity (ROC), survival analysis, etc.
- MCHP mapping tools (e.g., Geocoding Addresses (ArcView); Intra-urban Areas)
- Methods for making causal inferences:
- Propensity score matching [Methodology and SAS code (PDF)] - an alternative method to estimate the effect of receiving treatment when random assignment of treatments to subjects is not feasible.
- Instrumental variables [Introduction (PDF)] - used to control for confounding and measurement error, they allow for the possibility of making causal inferences with observational data.
- Study bias and validity both need to be carefully explored before and during the observational study:
- Validity - results should be checked against original or other sources for accuracy (within an acceptable percentage) and logic. Often, a gold standard [glossary term] is used. The most common gold standards are: patient chart reviews, a validated database, or survey data.
- Study bias - "can occur in any research and reflect the potential that the sample studied is not representative of the population it was drawn from and/or the population at large." (Mann 2003) Subject selection and loss-to-follow-up are examples of potential causes of study bias.
Publications about the Data Repository
The following is a list of publications that contain historical, descriptive information about the Manitoba Centre for Health Policy (MCHP) and the Manitoba Population Research Data Repository:
- Smith M, Roos L, Burchill C, Turner K, Towns D, Hong S, et al. Health Services Data: Managing the Data Warehouse: 25 Years of Experience at the Manitoba Centre for Health Policy. In: Sobolev B, Levy A, Goring S, editors. Data and Measures in Health Services Research. Boston, MA: Springer; 2015. p. 1–26. https://doi.org/10.1007/978-1-4939-8715-3_3
- Orr J, Smith M, Burchill C, Katz A, Fransoo R. Outcomes of an investment in administrative data infrastructure: An example of capacity building at the Manitoba Centre for Health Policy. (2016). Canadian Journal of Public Health, 107 (4-5), pp. e480-e481.
- Katz A, Brownell M, Enns J, Nickel N. Closing the loop: From system-based data to evidence-influenced policy and practice. In: International Journal of Population Data Science (IJPDS) Special Issue: Work designed to influence policy and practice. Vol.6 No. 3 (2021). https://doi.org/10.23889/ijpds.v7i1.1701
- Katz A, Enns J, Smith M, Burchill C, Turner K, Towns D. Population Data Centre Profile: The Manitoba Centre for Health Policy. In: International Journal of Population Data Science (IJPDS) Special Issue: Data Centre Profiles. Vol. 4 No. 2 (2019). https://doi.org/10.23889/ijpds.v4i2.1131
- Roos LL, Wall-Wieler E, Burchill C, Hamm NC, Hamad AF, Lix LM. Record linkage and big data—enhancing information and improving design. Journal of Clinical Epidemiology, Vol. 150 (2022), p 18-24. ISSN 0895-4356, https://doi.org/10.1016/j.jclinepi.2022.06.006.
https://www.sciencedirect.com/science/article/pii/S0895435622001524
Acknowledgements
The following guidelines/information on copyright, acknowledgement statements, and disclaimers are related to material published or presented that uses data from the Manitoba Population Research Data Repository at MCHP.
Copyright
Unless otherwise noted, all materials on the Manitoba Centre for Health Policy (MCHP) website are protected by copyright (©1995-2023)
All information contained within the Manitoba Centre for Health Policy's website is the property of MCHP and the University of Manitoba.
No part of the information on the MCHP website may be reproduced, stored, or transmitted in any form or by any means, without the written permission of MCHP, except as stated below:
MCHP grants permission to individuals to download or print information from our website for non-commercial, research, or personal purposes. Any person may reproduce short excerpts from the Centre's reports and publications for any purpose that respects the moral rights of authors, provided that the source is fully acknowledged.
As a courtesy, the consent of authors of such material should be obtained directly from the author.
Guideline
For an online acknowledgement, please use the following format that identifies the author(s), the page title, location (online address / URL) of the material, the last updated date, and the date of access. An example for an MCHP Concept Dictionary acknowledgement is provided below:
- Manitoba Centre for Health Policy. Concept: Asthma - Measuring Prevalence. Last Updated: 2017-03-07. Accessed April 19, 2017.
NOTE: Different publishers have different requirements, so use this as a guideline only.
Trademark information
The RDRG® system (formerly known as Refined Diagnosis Related Grouper) is licensed by MCHP and produced by Health Systems Consultants, Inc. The RGNs generated are based on a model developed by the Health Systems Management Group of Yale University under the Health Care Financing Administration (HCFA) Cooperative Agreements 15-C-98930/1-01 and 17-C-98930/1-0251. RDRG® is a registered trademark of HCFA.
Diagnosis Related Group (HCFA DRG) is a public domain grouper (“DRGFINDER® “) produced by 3M Health Information Systems under the Health Care Financing Administration Contract 500-94-0005. The HCFA DRGs produced by Health Systems Consultants, Inc should always be identical to those produced by 3M Health Information Systems.
CMGTM, PLxTM, & RIWTM are all registered trademarks of the Canadian Institute for Health Information (CIHI).
SAS® and all other SAS Institute Inc. product or service names are registered trademarks or trademarks of SAS Institute Inc. in the USA and other countries.
Adobe® and all related logos (e.g. Acrobat) are trademarks of Adobe Systems Incorporated.
Acknowledgement statements
Reports, publications, and presentations should include the following acknowledgment statements, if applicable:
1. MCHP Data Repository
Any publication should include the following acknowledgement:
The authors acknowledge the Manitoba Centre for Health Policy for use of data contained in the Manitoba Population Research Data Repository under project #___ (HIPC#___ or PHRPC#___). The results and conclusions are those of the authors and no official endorsement by the Manitoba Centre for Health Policy, Manitoba Health, or other data providers is intended or should be inferred. Data used in this study are from the Manitoba Population Research Data Repository housed at the Manitoba Centre for Health Policy, University of Manitoba and were derived from data provided by Manitoba Health and <name other data providers>.
For oral presentations, the acknowledgement can be done in one of three ways:
- Use MCHP template
- In describing the data used, indicate it is from the Manitoba Population Research Data Repository at MCHP. If the results are preliminary, say so (or something to indicate the study is ongoing); or
- Add the acknowledgement to a slide at the end of the presentation (likely not necessary since #2 is usually done anyway).
Please include Health Information Privacy Committee (HIPC) or the Provincial Health Research Privacy Committee (PHRPC) on the acknowledgements slide.
2. SAS®
Example statement: All data management, programming and analyses were performed using SAS® version ##.
Example wording for SAS (in publications)
The [output/code/data analysis] for this paper was generated using [SAS/STAT] software, Version [version number (e.g. 9.2)] of the SAS system for [Unix]. Copyright © [year of copyright] SAS Institute Inc. SAS and all other SAS Institute Inc. product or service names are registered trademarks or trademarks of SAS Institute Inc., Cary, NC, USA.
3. The Johns Hopkins ACG® Case-Mix System
Guideline: If you are using the ACG® system in your research please reference the software as "The Johns Hopkins Adjusted Clinical Group® (ACG®) Case-Mix System". Include the software version number used for the research and the most recent version number that is currently available as of the date of publication.
NOTE: The paper need not be about ACGs per se, nor is it necessary that ACGs be the central topic, but even if the system is used as a generic case-mix control in the background, the risk adjustment method or the markers utilized (say to identify chronic conditions) should be acknowledged.
NOTE: The current version of the system is version 11, installed in 2020. Version 10 was installed in 2014, Version 9 was installed in January 2011, and any work done prior to this used version 8.
Example statement: The use of Aggregated Diagnosis Groups™ (ADGs™) codes for risk adjustment in logistic models were created using The Johns Hopkins Adjusted Clinical Group® (ACG®) Case-Mix System version 11.
Note: Additional mentions of this system in the same document can simply refer to it as the ACG System or The Johns Hopkins ACG® Case-Mix System.
4. Census data
Guideline: If Canada Census data are used directly, (i.e. aside from creating income quintiles), please visit the Statistics Canada website. This website allows you to locate the particular set of Statistics Canada data that you've used and shows you how to cite it. You then use the reference to acknowledge them.
Example statement: Comparisons between XXX and census data were made using Statistics Canada 2001 census profile 20 per cent sample (Statistics Canada, 2004).
Example reference: Statistics Canada. 2006. Profile of the Population, 2006 Census. Statistics Canada Data Liberation Initiative Table Catalog no. 94-581-XCB2006002. Ottawa. July 24, 2008.
5. CHI Population Projection data
Guideline: If the Centre for Healthcare Innovation (CHI) Population Projection data are used in a project, they should be acknowledged as the developers of the methodology.
Example statement: The methodology used in the Population Projection data was developed by the Centre for Healthcare Innovation (CHI).
6. Use of Specific National Rehabilitation Reporting System (NRS) Data
Use of the Uniform Data Set, the FIM® Instrument and/or the impairment codes contained in the National Rehabilitation Reporting System (NRS) data, under the following circumstances, require acknowledgements that appear in the documentation. The following is a summary of the requirements. If necessary, the full requirements can be requested from MCHP.
- Insofar as any printed or published documentation refer to or incorporate any portion of the Uniform Data Systems for Medical Rehabilitation (UDSMR) materials, the following notice shall appear:
"The FIM® instrument, data set and impairment codes referenced herein are the property of Uniform Data Systems for Medical Rehabilitation, a division of UB Foundation Activities, Inc."
- Appropriate trade mark notices shall be incorporated into and appear in any printed or published documentation, and include the following notice:
“The service marks and trade-marks associated with the FIM® instrument are all owned by Uniform Data System for Medical Rehabilitation, a division of UB Foundation Activities, Inc. Use herein of service marks and trade-marks associated with the FIM® instrument is made with the permission of the Uniform Data System for Medical Rehabilitation.”
7. Territory acknowledgement
The following acknowledgement should be included in all presentations:
The University of Manitoba campuses are located on Treaty 1 land, the home of the Anishinaabeg, Cree, Oji-Cree, Dakota, and Dene peoples and on the homeland of the Red River Métis. We respect the Treaties that were made on these Territories, we acknowledge the harms and mistakes of the past, we recognize the ongoing present day colonial violence that is faced by Indigenous peoples within healthcare, education, justice, child welfare and government systems and we dedicate ourselves to moving forward in partnership towards decolonization in the spirit of reconciliation and collaboration.
Disclaimers
Although every effort has been made to ensure the information on our website is accurate, current, and reliable, MCHP assumes no responsibility or liability arising from any error, omission, opinions expressed by authors, or from the use of any information contained herein.
Medical definitions and descriptions
Any descriptions of diagnoses, procedures, and tests are provided for educational and informative purposes only. This information should not be used for diagnosing or treating a health problem or a disease, and it is not a substitute for professional medical care. If you have, or suspect you have, a medical condition, you should consult your own primary health care physician for further information and advice.
While all efforts have been made to ensure the accuracy of this information, neither MCHP nor the University of Manitoba can be held responsible for its accuracy, nor for any actions made on the basis of information provided on MCHP’s website.
Links to external sites
This disclaimer is extended to the content of external websites to which we have created links. MCHP has no control over the availability of remote sites, and is not responsible for the information found at those sites. The inclusion of a link on our website does not constitute an endorsement by MCHP or the University of Manitoba.
Health Data Research Network Canada (HDRN Canada)
Health Data Research Network Canada (HDRN Canada)
The Manitoba Centre for Health Policy (MCHP) is a member of Health Data Research Network Canada (HDRN Canada), a pan-Canadian group of organizations that aims to improve health and well-being by making multi-jurisdictional data more accessible to researchers, institutions and government agencies across Canada for research.
HDRN Canada connects individuals and organizations from across the country to share expertise, identify opportunities for collaboration, and foster innovation in ways that respect public expectations and Indigenous data sovereignty.
The SPOR Canadian Data Platform (SPOR CDP) is the first initiative of HDRN Canada. In 2018, HDRN Canada was awarded a $81.35M, seven-year grant from the Canadian Institutes of Health Research (CIHR) to develop the SPOR CDP.
SPOR CDP infrastructure supports improved access to data, automation of data analysis, and ongoing engagement with the public, patients, and Indigenous communities.
In 2020, HDRN Canada launched the Data Access Support Hub (DASH), a one-stop data access service portal where researchers can request access to data from multiple provincial, territorial and pan-Canadian sources, and track the progress of their application.
For more information, please visit: https://www.hdrn.ca/
Centre for Healthcare Innovation (CHI)
The services provided by the George and Fay Yee Centre for Healthcare Innovation (CHI) include:
- clinical research and methods
- data analytics and management (e.g. use of REDCap for collecting and storing project specific data)
- a learning health system
- patient and public engagement, and
- patient reported measurement
For more information, see CHI Services or contact CHI.
To request a free consultation, please complete and submit a:
CHI and MCHP Request for Service form
Contact us
Manitoba Centre for Health Policy
Community Health Sciences, Max Rady College of Medicine
Rady Faculty of Health Sciences,
Room 408-727 McDermot Ave.
University of Manitoba
Winnipeg, MB R3E 3P5 Canada
Charles Burchill
Associate Director, Data and Use,
Manitoba Population Research Data Repository
