Richard Keijzer
Associate dean (Research)
204-789-3375
richard.keijzer@umanitoba.ca
Max Rady College of Medicine
Research and scholarly activity
Improving health through education, research and partnerships. The Max Rady College of Medicine is a leader in the areas of immunity, infectious disease, population and global health, patient-oriented research and integrative research in health and well-being.
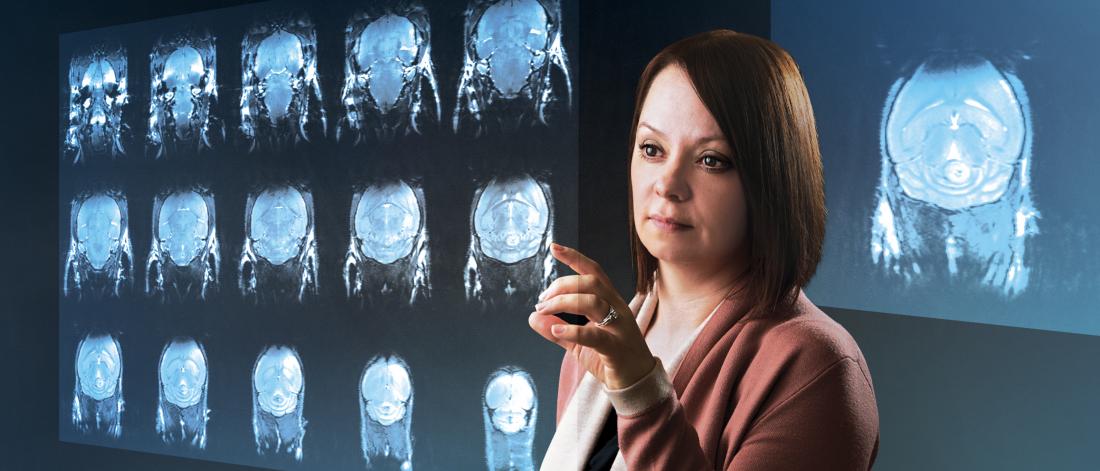
Research and scholarly activity
Areas of expertise
Researchers in our health-focused member colleges are individually recognized as experts in their field, leading discovery in a broad range of interests that have an impact that can be seen everywhere in our daily lives.
Department research
Learn more about our department-level research priorities and see where our research is leading.
Clinical departments
Supporting research
The Max Rady College of Medicine is committed to funding and supporting research. The resources below will connect you with people, spaces, equipment, opportunities and policy or guideline-related information — all in effort to enhance your journey to discovery.
You may also be looking for
-
Apr
22Alan Klass Program in Health Humanities: Creative and Reflective Writing as a Mindfulness Practice
12:00 PM
Online Zoom Session -
Apr
24adMare Academy BioInnovation Scientist Program: Industry Essentials
12:00 AM
-
Apr
24Praxis Series: Identifying and Addressing Inadequacies in Musculoskeletal Medical Education Across Canada
11:00 AM
Online Zoom Session
Associate Dean (Research) Contact us
-
-
Carla Dunton
Office assistant
204-789-3375
carla.dunton@umanitoba.ca -
Barbie Haddon
Administrative assistant
204-789-3886
barbie.haddon@umanitoba.ca
Our office
Office of the Associate Dean (Research)
A203 Chown Building
753 McDermot Avenue (Bannatyne campus)
Winnipeg, MB R3E 0T6 Canada








