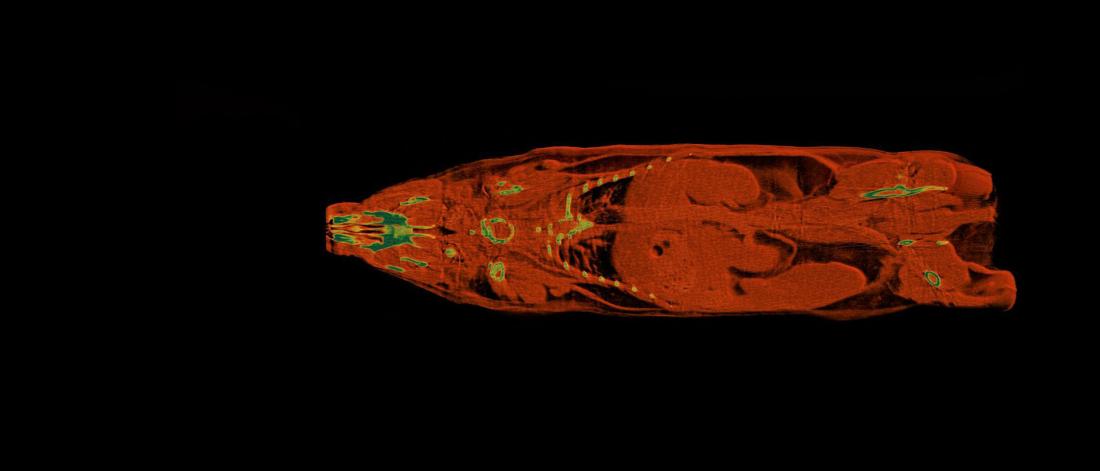
Central Animal Core Imaging and Transgenic Facilities
Forms
Use the links below for quick access to our forms.
Fees
Costs for our services are listed here.
Transgenic services and surgeries
These prices are for internal investigators. External academic and industry clients should contact us for pricing.
| Item | Cost | Notes |
|---|---|---|
| Cell line preservation | ||
| Embryo collection/harvesting E0.5-3.5 | $150 | per mouse |
| Embryo collection/harvesting E9.5-17.5 | $75 | per mouse |
| ES cell derivation | Please contact us for pricing/availability | per attempt |
| Mouse embryonic fibroblasts | $750 | per pregnant mouse, if genotype known |
| Mouse embryonic fibroblasts | $1,000 | per pregnant mouse, to generate two different lines |
| Chimera production | ||
| Aggregation | Please contact us for pricing/availability | |
| Blasotcyst injection | Please contact us for pricing/availability | |
| Crispr/Cas9 conditional model | $9,750 | Rady Faculty of Health Sciences primary appointments |
| Crispr/Cas9 conditional model | $13,000 | All others |
| Crispr/Cas9 knockout or knockin | $4,875 | Rady Faculty of Health Sciences primary appointments |
| Crispr/Cas9 knockout or knockin | $6,500 | All others |
| Pronuclear injection | Please contact us for pricing/availability | |
| Cryopreservation and recovery | ||
| Ovary freezing | $500 | per attempt |
| Speed embryo cryopreservation and sperm freezing | $1,800 | per line |
| Speed embryo cryopreservation | $1,500 | per line |
| Sperm freezing | $500 | per attempt, with motility test |
| Traditional embryo cryopreservation | $1,500 | per attempt for up to three sessions. Additional charges may apply if more sessions are required to obtain suitable numbers of embryos for storage |
| Gene Targeting | ||
| Electroporation/selection | $2,000 | per attempt |
| Sequencing | $15 | per sample: DNA extraction, PCR, purification, sending for Sanger sequencing and analysis |
| Vector construction | Please contact us for pricing/availability | |
| Microinjection | $2,000 | per attempt |
| Rederivation | ||
| Embryo rederivation with transfer into CD1 pseudo-pregnant female | $750 | per attempt |
| Sperm rederivation with in vitro fertilization followed by embryo transfer | $1,750 | per attempt |
| Storage | ||
| Ovary storage | $300 | per year, first line |
| Sperm storage | $300 | per year, first line |
| Additional line | $50 | per year |
| Other animal services | ||
| Necropsy | $50 | per mouse |
| Ovariectomy | $75 | per mouse |
| Vasectomy | $75 | per mouse |
| Genotyping | $8 | per sample: DNA extraction, single PCR and Polyacrylamide gel |
| Miscellaneous services | $50 | technician time rate per hour |
| Other services | Please contact us | |
Imaging
These prices are for internal investigators. External academic and industry clients should contact us for pricing.
| Academic | Academic | Industrial | |
|---|---|---|---|
| Instrumentation | CACITF operated (per hour) | PI operated (per hour) | CACITF operated (per hour) |
| IVIS Spectrum | $125 | $75 | $250 |
| Micro CT | $125 | $75 | $250 |
| PET-MRI | $125 | $75 | $250 |
| Micro PET | $125 | $75 | $250 |
| Ultrasound | $125 | $75 | $250 |
| SPECT-CT | $125 | $75 | $250 |
Prices include supplies such as syringes, anesthetic etc. Fees do not include reagents such as fluorescence and bioluminescent probes, vascular contrast agents or radioisotopes.
Animal surgeries and procedures
Central Animal Core Imaging and Transgenic Facilities can facilitate a wide variety of surgical and non-surgical animal services for researchers working within our facilities.
Necropsy
Necropsies on mice are performed for a number of reasons, including harvesting of tissues for research, health surveillance and investigation of disease.
What we will do
Isoflurane anesthesia is induced, followed by tissue collection.
Tissues are then placed in fixative or frozen as required by the investigator. Standard tissues collected include brain, heart, liver, kidney, lungs, spleen, testes (or ovary/uterus), skeletal muscle, but other tissues may be requested by the investigator.
What you need to supply
- Mouse to be necropsied
Special considerations
The mouse is sacrificed during this procedure.
Fees
Please see the fees section for details.
Non-invasive blood pressure measurement
The IITC MRBP tail cuff blood pressure measurement system offers a non-invasive way to to monitor the blood pressure of mice and rats.
What we will do
The MRBP system consists of climate controlled restraint chambers in which up to six animals may be placed. The animals are fitted with a combined inflation-detection cuff at a user-selected ambient temperature (typically 32 Celcius or lower).
Blood pressure pulses are detected by a high sensitivity photoelectric sensor. Readings are monitored, recorded and stored automatically and can be exported into an Excel spreadsheet.
What you need to supply
- Up to six rats or mice
Fees
Please see the fees section for details.
Ovariectomy
Performing an ovariectomy on a female mouse produces a sterile animal, allowing investigators to study hormones and their effects.
What we will do
We will perform the surgery and observe the mice during recovery for up to two weeks.
What you need to supply
Researchers must provide:
- Female mouse to be ovariectomized
Special considerations
The investigator is responsible for all housing costs.
Fees
Please see the fees section for details.
Vasectomy
Vasectomies are performed on male mice to produce sterile males.
What we will do
The vas deferens is cut or cauterized such that reconnection is unlikely. Following surgery, males should be allowed a recovery period of two weeks, during which the surgical clips are removed.
Vasectomized males are test-mated with two females to confirm the success of the vasectomy. Pregnancy is normally apparent within two weeks. If not visible after this time, the male is considered suitable for use.
What you need to supply
Investigators must provide:
• Male mouse to be vasectomized
• Two female mice for test breeding
Special considerations
The facility will perform the surgery, but the costs of the two female mice for breeding, and the boarding costs of the male and female mice are the responsibility of the investigator.
Fees
Please see the fees section for details.
Transgenic services
We provide comprehensive transgenic services for mice to support research by University of Manitoba investigators and external clients.

Cell line generation and preservation
Producing a new mouse model takes significant resources. Protect your investment by generating cell lines for in vitro studies or as the basis for further genetic modification.
Generation of mouse embryonic stem cells
Mouse embryonic stem cells (mESCs) are an important tool for the investigation of pluripotency and lineage analysis.
Deriving mouse embryonic stem cells (mESCs) from genetic models provides an important tool for the analysis of the effect of a genetic change on development. It can also provide a tool for additional genetic manipulation.
What we will do
Embryos will be collected from the model of interest at E3.5, and at E4.5, epiblasts will be collected from late stage blastocysts and transferred to culture.
Each embryo will be treated separately and a small portion of successfully derived lines will be provided to the investigator for genotyping.
Up to 4 ES cell lines will be expanded and three vials of each will be frozen and supplied to the investigator.
What you need to supply
- Three female mouse for breeding (and the mutant breeder male).
- Demonstration that an assay is in place for genotyping.
Special considerations
The investigator will need to perform the genotyping within 24 hours of receiving samples.
Fees
Please see the fees section for details.
Mouse embryonic fibroblasts
Creating Mouse Embryonic Fibroblasts (MEFs) allows investigators to establish a primary culture from genetically modified animals.
What we will do
MEFs are generated from E13.5 or E14.5 dcp (day post-coitum) embryos. Cells may be obtained from an earlier stage of development if homozygosity of a line is lethal before E13.5, although the yield of fibroblasts will be reduced.
MEFs are then expanded and frozen in liquid nitrogen indefinitely for future use.
Known genotypes
If the embryos are of known genotype (i.e. both parents are homozygous at the locus of interest), we will extract MEFs from up to three embryos, culture and harvest the cells. Up to five aliquots of 3 x 106 per cryo-vial will be frozen. A “thaw test” will be performed before transferring the cells to the requestor.
Unknown genotypes
If the embryos are of unknown genotype (i.e. parents are heterozygous), we will extract MEFs from up to 10 embryos. The requester will be asked to genotype a small sample from each embryo within 24 hours, and we will continue to culture up to three embryos of each desired genotype.
What you need to supply
- Female mouse/mice
- Male breeder from which to establish a MEF cell line
Special considerations
The animals used for this procedure will be assigned to the facility transgenic protocol, so investigators do not need to write a protocol for this procedure or to cover these animal numbers.
Fees
Please see the fees section for details.
Embryo collection
Harvesting of embryos can be performed for a variety of applications.
What we will do
Embryos will be collected from the timed-pregnant mice and treated as requested by the investigator. The embryos can be collected at different stages from timed-pregnant female mice.
What you need to supply
- Female mice for generating timed-pregnancies
- Solutions and instructions for embryo processing
- An animal protocol for the timed pregnant females
Special considerations
Investigators should be aware that the number of embryos present in a timed pregnant mouse is highly variable.
Fees
Please see the fees section for details.
Cryopreservation and recovery
Cryopreservation is a cost-effective way to preserve rare or unique mouse strains. It can also help protect against losses due to disease outbreaks in animal facilities.
Embryo freezing
Embryo freezing allows investigators to permanently store mouse lines.
The investigator should be aware that the number of embryos that can be collected from these methods varies significantly among strains. Although the facility attempts to store at least 100 embryos per line, this cannot be guaranteed.
Fees
Please see the fees section for details.
Rederivation from fresh/frozen embryos
What we will do
Prepare medium for thawing of the embryos based on the protocol supplied by the site that froze the embryos.
Thawed embryos will be transferred to pseudopregnant females.
The mice will be monitored until delivery and samples from the offspring will be provided at weaning to the investigator for genotyping
Pups with the desired genotype will be transferred to the investigator’s protocol.
What you need to supply
- Frozen embryos or female/male mice to use to generate embryos
- Confirmation of the availability of an assay to genotype the offspring
- An animal protocol to accept the offspring
Special considerations
The facility cannot guarantee the success of an embryo transfer. If the investigator is ordering embryos from an external source, assuring that a control embryo vial is provided by the vendor can increase the likelihood of success. The control embryos will be used to optimize conditions.
Fees
Please see the fees section for details.
Ovary freezing
Ovary freezing allows investigators to permanently store mouse lines. It also allows investigators to “rescue” transgenic lines that are difficult to propagate.
What we will do
When the ovaries are collected, they are cut in half and either frozen in a cryovial or transferred to a histocompatible female.
If the ovary is transferred to another female, the mouse will be transferred to the investigator for testing the viability of the transferred ovary.
The animal protocol for the mice and procedure, as well as testing of the ovary viability, is covered by the Central Animal Care breeding protocol until successful transmission of the genetic alteration of interest is demonstrated.
At that time, the investigator will need to obtain an approved animal protocol.
What you need to supply
- Female mice at four to eight weeks of age from which to generate the ovaries.
- Female mice for implanting the ovaries (histocompatible female mice).
Storage
The frozen ovaries can be transferred to the investigator for storage or stored in the facility.
Special considerations
- Female donor mice will be sacrificed.
- The facility cannot guarantee the success of the ovary transplantation.
- Investigators are responsible for all housing costs.
Fees
Please see the fees section for details.
Sperm freezing
Sperm freezing allows investigators to permanently store mouse lines.
What we will do
Sperm is collected from male mice demonstrated to be fertile. Sperm will be collected and frozen in aliquots of 150 ul x 6 cryovials. One vial will be thawed and the sperm motility tested.
What you need to supply
- Two proven-fertile male mice.
Storage
The frozen sperm may be transferred to the investigator for storage or stored in the Facility (in two different storage places).
Special considerations
The male mouse is sacrificed for the collection of sperm.
Fees
Please see the fees section for details.
Rederivation from fresh/frozen sperm
What we will do
Prepare medium for thawing of the sperm based on the protocol supplied by the provider.
Fresh or frozen mouse sperm will be used for in vitro fertilization of mouse oocytes collected from superovulated female mice. Fertilized oocytes will be transferred to pseudopregnant females.
The mice will be monitored until delivery and samples from the offspring will be provided at weaning to the investigator for genotyping. Pups with the desired genotype will be transferred to the investigator’s protocol.
What you need to supply
- Frozen sperm or male mice that are proven to be fertile (for sperm collection)
- Confirmation of the availability of an assay to genotype the offspring
- An animal protocol to accept the offspring
Special considerations
The facility cannot guarantee the success of an in vitro fertilization. If the investigator is ordering sperm from an external source, assuring that a control sperm sample is provided by the vendor is important to increase the likelihood of success. This control sperm sample will be used to optimize the procedure.
Fees
Please see the fees section for details.
Mouse model generation
CRISPR/Cas9 is now the method of choice for the generation of most mouse models. Our facility has successfully used CRISPR/Cas9 to generate knock-in, knock-out and conditional (i.e. floxed) mouse alleles.
Aggregation
Diploid embryo/ES cell aggregation can generate a mouse chimeric for a genetic alteration of interest.
What we will do
Genetically modified mouse embryonic stem cells (mESCs) are aggregated with diploid CD1 embryos to generate chimeric mice. The ES cell line will be expanded and the cells will be aggregated with a minimum of 150 CD1 embryos.
The aggregated embryos will be transferred into CD1 pseudo-pregnant females.
Offspring will be monitored for chimeras. At weaning, chimeras can either be transferred to the investigator’s protocol for testing of germ line transmission or maintained for up to two weeks at the investigator’s expense.
What you need to supply
- The ES cell line with evidence of mycoplasma testing (chromosome counting is also recommended)
- Evidence that a screening procedure has been planned to identify the transgene
- An animal protocol is required for the transfer of the founding mice
Special considerations
Please note that the facility cannot guarantee germline transmission of different mouse ES cells and chimeras.
Fees
Please see the fees section for details.
Blastocyst injection
Blastocyst injection of genetically altered mouse ES cells is used to generate chimeric mice carrying the genetic alteration(s).
What we will do
ES cells will be expanded for microinjection and injected into 20-30 albino B6 blastocysts. They will then be transferred into two to three CD1 pseudo-pregnant females.
Offspring will be monitored for chimeras and at weaning, transferred to the investigator for testing of germ line transmission. They can also be maintained for up to two weeks at the investigator’s expense.
What you need to supply
- The ES cell line with evidence of mycoplasma testing (chromosome counting is also recommended)
- Evidence that a screening procedure has been planned to identify the transgene
- An animal protocol is required as soon as there is evidence that germline transmission has been obtained
Special considerations
Please note that the facility cannot guarantee germline transmission of different mouse ES cells and chimeras.
Fees
Please see the fees section for details.
CRISPR/Cas9
CRISPR/Cas9 allows precise edits to be made in order to create knock-out, knock-in or conditional mouse models.
What we will do
After meeting with the investigator, a design for the model will be created.
Once the design has been approved by the investigator, the reagents (guide RNA and ss Donor if applicable) will be ordered.
The editing efficiency of the reagents will be tested in zygotes prepared by in vitro fertilization. If editing efficiency is acceptable, zygotes will be electorporated and transferred to pseudopregnant female mice.
After weaning, the founders will be transferred to the investigator's protocol or maintained for up to two weeks at the investigator's expense.
What you need to supply
Female and male mice will need to be provided if the gene edits are to be made on a mutant background. The facility will supply wild type C57BL6 or CD1 mice.
Special considerations
The investigator must prepare an animal protocol to receive the founder mice. The facility cannot guarantee that the targeted gene will be successfully altered or transmitted in the germ line.
Fees
Please see the fees section for details.
Gene targeting
Gene targeting vectors or expression vectors can be introduced into ES cells by electroporation and selected for subsequent screening and aggregation.
Most gene targeting is now done using CRISPR/Cas9. For specialized models that require vector construction please contact us directly to discuss the approach and pricing.
What we will do
We will meet with the investigator to determine the scope of the project. Then, strategy and workflow will be planned individually for each project.
What you need to supply
The role of the facility and investigator will be optimized for each project.
Special considerations
This approach is no longer used routinely by our facility as this approach has been largely replaced by CRISPR/Cas9 mediated modification of mouse genes.
Fees
Please see the fees section for details.
Pronuclear injection
Pronuclear injection allows for the introduction of DNA or RNA for expression in the genome.
What we will do
Pronuclear injection is commonly used to inject genetic material directly into the nucleus of a fertilized oocyte. Fertilized eggs are collected at E0.5 when they are a single cell. Linearized DNA, RNA and/or protein, as appropriate, is injected into one of the pronuclei. The injected oocytes are then transferred into a pseudo pregnant mouse.
We will inject the DNA into a minimum of minimum of 150 oocytes and implant them back into female mice.
Founders (minimum of three) will be monitored. At three weeks, tail biopsies will be obtained and provided to the investigator for genotyping.
After weaning, the founders will be transferred to the investigator for testing germline transmission.
Before you begin
Before contacting us, please have the following in place:
- Justification to generate the model
- An animal protocol to receive the founder mice
- A genotyping assay for the transgene
What you need to supply
- DNA for injection. Please consult the facility for a protocol for preparation of the DNA for injection. If required, the facility can prepare, quantify, and digest the construct, and prepare the insert for injection.
- Three ug DNA at 100 ng/ul. We will dilute this in injection buffer to a concentration of 50 ng/ul.
- A picture verifying the quality, quantity and complete digestion of the DNA to be injected.
Special considerations
Please note that the facility cannot guarantee that the transgene will be expressed or transmitted in the germ line.
Fees
Please see the fees section for details.
Citing UM transgenic services
Citing the services you received acknowledges the contributions of the transgenic services, central animal core (CAC) staff and resources for animal studies performed at UM facilities.
You can use the methods or acknowledgments section in your poster, meeting presentations and published work.
When significant intellectual contributions have been made by the UM transgenic service team and CAC staff, please follow the accepted scientific practice for publication. When appropriate, the recognition of UM team members as co-authors is appreciated.
The tabs below list publications that involve the use of transgenic services or CAC staff from the UM. For a complete publications list (ranging from 2023 to 2003) please submit a request to transgenicservice@umanitoba.ca.
If you would like to submit references of publications or patents in which the services of UM transgenic facilities were used to generate models used in the published studies, please email transgenicservice@umanitoba.ca — Pubmed IDs are sufficient.
US Patents on mouse models
1. Various uses of obesity-linked cancer models. Canada. US Patent # 62/234,336.
2. Mito-Ob: A transgenic mouse model for obesity. Canada. US20150026833 A1.
2023
Wu X, Simard LR, Ding H. Generation of Conditional Knockout Alleles for PRUNE-1. Cells. 2023 Feb 6;12(4):524. doi: 10.3390/cells12040524. PMID: 36831191; PMCID: PMC9954577.
2022
Bassi G, Mishra S. Prohibitin-1 plays a regulatory role in Leydig cell steroidogenesis. iScience. 2022 Mar 28;25(4):104165. doi: 10.1016/j.isci.2022.104165. PMID: 35434552; PMCID: PMC9010647.
Xu YXZ, Ande SR, Ikeogu NM, Zhou K, Uzonna JE, Mishra S. Prohibitin plays a role in the functional plasticity of macrophages. Mol Immunol. 2022 Apr;144:152-165. doi: 10.1016/j.molimm.2022.02.014. Epub 2022 Feb 24. PMID: 35219912.
2019
Xu YXZ, Bassi G, Mishra S. Prohibitin: a prime candidate for a pleiotropic effector that mediates sex differences in obesity, insulin resistance, and metabolic dysregulation. Biol Sex Differ. 2019 May 22;10(1):25. doi: 10.1186/s13293-019-0239-5. PMID: 31118075; PMCID: PMC6530082.
Mishra S, Nyomba BG. Prohibitin: A hypothetical target for sex-based new therapeutics for metabolic and immune diseases. Exp Biol Med (Maywood). 2019 Feb;244(2):157-170. doi: 10.1177/1535370219828362. Epub 2019 Feb 4. PMID: 30717609; PMCID: PMC6405819.
2018
Xu YXZ, Mishra S. Obesity-Linked Cancers: Current Knowledge, Challenges and Limitations in Mechanistic Studies and Rodent Models. Cancers (Basel). 2018 Dec 18;10(12):523. doi: 10.3390/cancers10120523. PMID: 30567335; PMCID: PMC6316427.
Xu YXZ, Ande SR, Mishra S. Gonadectomy in Mito-Ob mice revealed a sex-dimorphic relationship between prohibitin and sex steroids in adipose tissue biology and glucose homeostasis. Biol Sex Differ. 2018 Aug 29;9(1):37. doi: 10.1186/s13293-018-0196-4. PMID: 30157935; PMCID: PMC6114179.
Imaging
Through our unit, advanced imaging systems are available to support academic, preclinical and industrial research involving in vivo studies of small animals and ex vivo studies of materials (mineralized biomaterials, grafts, implants, composites etc.). Where possible, we provide data acquisition and analysis training using our instrumentation.
-
We also offer support with:
- Budgets
- Ethics submissions
- Experimental design
- Feasibility assessments
- Grant and manuscript preparation
Video recording
Overview
CACITF offers video recording services for both animal- and non-animal related research. This service provides the opportunity to record your work for instructional purposes or as material for video journals.
Equipment
Our equipment includes:
- ATEM Mini Pro Stream Switcher
- Black Magic Pocket Cinema Camera 4K video cameras
- High definition (4K) video cameras
- Laptop
- Lenses
- Lumix G X Vario 12-35mm f/2.8 II ASPH Power OIS Lens
- Microphones
- Monitor
- MT190X3 Tripod + 556B Levelling Column + MVH500AH Head
- Portable lavalier set w/(1) ME2-II clip-on lapel mic, (1) XSW-D MINI Jack TX (3.5mm), (1) XSW-D MIN
- Tripods
- Video switcher
- All required cables
The ATEM switching unit allows up to four devices (cameras, laptops etc) to be connected via HDMI input ports. Video can be output to a display monitor, an external hard drive and live streamed (via Skype, Zoom, Teams or directly via internet connection). All four inputs plus the output are displayed on the output monitor. The input to be output to video is selected simply by pushing a button, as is adding switching transition effects and saving video.
Booking
Any investigator based at the University of Manitoba may request access to this service using our request form.
Please note that video recording of any research involving animals must first be approved by the University of Manitoba Animal Care Committee via an amendment. Once your amendment is approved, enquiries/requests can be submitted online.
Request video recording services
Fees
CACITF offers video recording services at $250 per half-day or $500 for a full day.
If you do not need assistance, equipment may be used at $50 per half-day or $100 for a full day.
Training and reservations
Image training and account request form
All imaging equipment users must have received appropriate training and have an account to be able to reserve equipment. To arrange training and obtain a reservation account please complete the training and account request form.
Reservations
Trained investigators can use our booking form to reserve equipment.
You may also be looking for
Contact us
Central Animal Core Imaging and Transgenic Facilities
23 Basic Medical Sciences Building
745 Bannatyne Avenue
University of Manitoba
Winnipeg, MB R3E 0J9 Canada
Transgenic services: transgenicservice@umanitoba.ca
Imaging services: SAMICF@umanitoba.ca
